Anti-Infliximab (Remsima®)ADA Semi-Quantitative ELISA Kit
- SKU:
- HUMB00008
- Product Type:
- ELISA Kit
- ELISA Type:
- Biosimilar ELISA
- Biosimilar ELISA Type:
- Total Semi-quantitative
- Applications:
- ELISA
- Reactivity:
- Human
- Analytes:
- Infliximab (Remsima®)
- Research Area:
- Anti-TNF Alpha
Description
Anti-Infliximab ADA Semi-Quantitative ELISA Kit
Enzyme-linked immunosorbent assay for the semi-quantitative determination (screening) of total antibodies to infliximab biosimilar (Remsima®) in serum and plasma. Remsima® is the world's first biosimilar mAb (approved in 2013 by the EMA). The demonstration of anti-infliximab antibodies during treatment with infliximab (Remsima®) is a major concern and monitoring for the presence and/or quantitation of specific antibodies is vital. With the Assay Genie Anti-Infliximab ADA Semi-Quantitative ELISA Kit, infliximab anti-drug antibodies (ADA) that are bound to infliximab in serum, that can't be detected by free antibody detection kits can be determined. This Assay Genie ELISA kit is for research use only.
Anti-Infliximab (Remsima®) ADA Semi-Quantitative ELISA Kit test principle
The Assay Genie Antibody to infliximab (Remsima®) ELISA is a sandwich assay for the determination of total antibodies against infliximab in serum and plasma samples. During the first incubation period, the separation of infliximab specific antibodies is provided by adding dissociation buffer. After transferring dissociation mix to the plate, anti-infliximab antibodies are separated from infliximab in patient serum/ plasma samples. These are captured by the drug infliximab (Remsima®) coated on the wall of the microtiter wells and peroxidase-labelled specific conjugate. After washing away the unbound components from samples, the bound enzymatic activity is detected by the addition of tetramethylbenzidine (TMB) chromogen-substrate. Finally, the reaction is terminated with an acidic stop solution. The intensity of the reaction colour is directly proportional to the concentration of anti-infliximab antibodies in the sample.
Anti-Infliximab (Remsima®) ADA Semi-Quantitative ELISA Product Information
| Information | Description |
Application | Free drug |
Required Volume (uL) | 10 |
Total Time (min) | 95 |
Sample Type | Serum, Plasma |
Number of Assays | 96 |
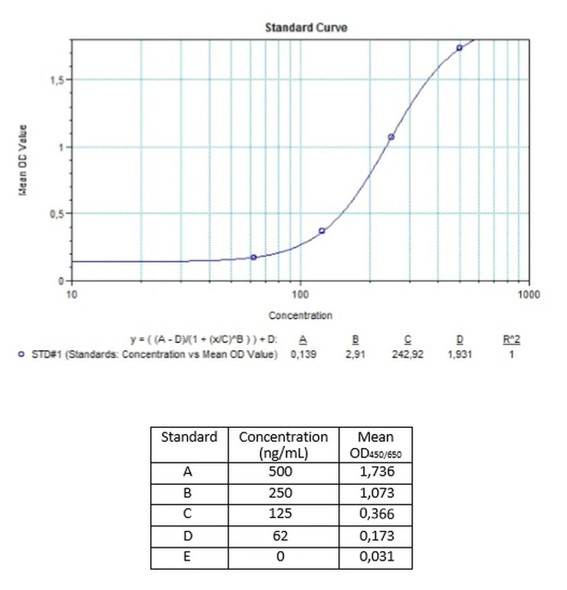
Detection Limit (ng/mL) | 156 (ng/mL) |
Spike Recovery (%) | - |
Shelf Life (year) | 1 |
Alternative Names | Tumour Necrosis Factor Alpha Remsima |
Anti-Infliximab ADA Semi-Quantitative - Key Information
Infliximab mode of action
Infliximab (Remsima®) is a chimeric monoclonal antibody and used to treat autoimmune disorders. Infliximab reduces the amount of active human tumour necrosis factor alpha (hTNF alpha) in the body by binding to it and preventing it from signalling the receptors for TNF alpha on the surface of various cell types. TNF alpha is one of the key cytokines that triggers and sustains the inflammatory reactions.
Infliximab uses
Remsima is the world’s first biosimilar mAb (approved in 2013 by the EMA). The Committee for Medicinal Products for Human Use (CHMP) decided that, in accordance with EU requirements, Remsima was shown to have a comparable quality, safety and efficacy profile to Remicade. Remsima is a tumor necrosis factor alpha (TNF-alpha ) antagonist used to treat rheumatoid arthritis, ankylosing spondylitis, ulcerative colitis, adult Crohn's disease, plaque psoriasis, and psoriatic arthritis.
Infliximab immunogenicity
One of the major concern, despite of its wide usage, is potential development of anti-infliximab antibodies which in turn may interfere with infliximab (Remsima®) efficacy as primarily determined by observing the return of signs and symptoms of disease. This could lead to therapeutic dose-escalation or a potential termination of treatment. The demonstration of anti-infliximab antibodies during treatment with infliximab (Remsima®) is a major concern and monitoring for the presence and/or quantitation of specific antibodies during clinical trials is important for follow up of the treatment regimens.
With the Assay Genie Anti-Infliximab (Remsima®) ADA Semi-Quantitative ELISA Kit, infiliximab-specific antibodies that are bound to infiliximab in serum that can’t be detected by free antibody detection kits can be determined in patients receiving Remsima®. This Assay Genie ELISA test can be efficiently used for monitoring infliximab-specific antibodies and is for research use only.
Anti-Infliximab ADA Semi-Quantitative ELISA Kit Contents
| Size | Kit Contents |
1 x 12 x 8 | Microtiter Plate Break apart strips. Microtiter plate with 12 rows each of 8 wells coated with infliximab |
1 x 0.25 mL | Reactive Control |
1 x 0.5 mL | Negative Control |
1 x 0.25 mL | Immune Complex Control |
1 x 12 mL | Dissociation Buffer |
1 x 6mL | Neutralisation Buffer |
1 x 50 mL | Assay Buffer |
1 x 12 mL | Peroxidase-Conjugate. Red coloured. Ready to use. Contains peroxidase (POD) conjugate, stabilizer and preservatives. |
1 x 12 mL | TMB Substrate Solution |
1 x 12 mL | TMB Stop Solution |
1 x 50 mL | Wash Buffer concentrate (20x) |
2 x 1 | Adhesive Film |
1 x 12 x 8 | Microtiter plate Break apart strips. Microtiter plate for dilution |
Anti-Infliximab ADA Semi-Quantitative ELISA Protocol
| Steps | Protocol |
1 | Pipette 10μL of ready-to use Low Control, Reactive Control, and Samples into the respective wells of dilution plate. |
2 | Add 90μL dissociation buffer to dilution plate and incubate plate 15 min at room |
3 | After 15 min incubation, pipette 65μl of Peroxidase conjugate and 35μl |
4 | Cover the plate with adhesive film. Briefly mix contents by gently shaking the plate. Incubate 60 min at room temperature (18-25°C). |
5 | Remove adhesive film. Discard incubation solution. Wash plate 3 times each with 300μL of diluted Wash Buffer. Remove excess solution by tapping the inverted plate on a paper towel. |
6 | Pipette 100μL of TMB Substrate Solution into each well and incubate 20 min (without adhesive foil.) at room temperature (18-25°C) in the dark. |
7 | Stop the substrate reaction by adding 100μL of Stop Solution into each well. Briefly mix contents by gently shaking the plate. Colour changes from blue to yellow |
8 | Measure optical density with a photometer at 450/650 nm within 30 min after pipetting of the Stop Solution. |
Trademarks
REMSIMA is a trademark of CELLTRION, INC.