Enzyme-linked immunosorbent assay for the quantitative determination of free Cetuximab (Erbitux®) in serum and plasma. The Assay Genie Cetuximab ELISA has been especially developed for the quantitative determination of free Cetuximab in serum and plasma samples and is for research use only.
Description
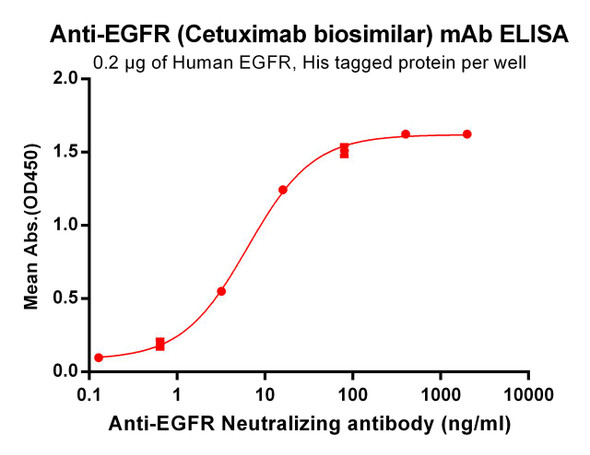
Cetuximab ELISA Kit
Cetuximab (Erbitux®) ELISA Kit test principle
Solid phase enzyme-linked immunosorbent assay (ELISA) based on the sandwich principle. Standards and samples (serum or plasma) are incubated in the microtitre plate coated with the reactant for cetuximab (Erbitux®). After incubation, the wells are washed. A horse radish peroxidase (HRP) conjugated probe is added and binds to cetuximab (Erbitux®) captured by the reactant on the surface of the wells. Following incubation wells are washed and the bound enzymatic activity is detected by addition of chromogen-substrate. The colour developed is proportional to the amount of cetuximab (Erbitux®) in the sample or standard. Results of samples can be determined directly using the standard curve.
Cetuximab (Erbitux®) ELISA Product Information
| Information | Description |
| Application | Free drug |
| Required Volume (μl) | 10 |
| Total Time (min) | 70 |
| Sample Type | Serum, Plasma |
| Number of Assays | 96 |
| Detection Limit (ng/mL) | 100 (ng/mL) |
| Spike Recovery (%) | 85-115% |
| Shelf Life (year) | 1 |
| Alternative Names | Anti-Epidermal Growth Factor Receptor (EGFR) mAb Erbitux |
Cetuximab (Erbitux®) - Key Information
Cetuximab (Erbitux®) mode of action
Cetuximab (IMC-C225, Erbitux) is a human/murine chimeric monoclonal antibody of the immunoglobulin G1 (IgG1) and FDA-approved epidermal growth factor receptor (EGFR) inhibitor. It is a 152 kDa protein composed of four polypeptide chain. There are 32 cysteine residues forming accordingly 16 potential disulfide bonds.
Cetuximab (ICM-225, Erbitux™) works by binding to the extracellular domain III of EGFR. This binding partially blocks the EGFR ligand-binding domain and sterically hinders the correct extended conformation of the dimerization arm on domain II. Cetuximab therefore prevents both ligand binding and the proper exposure of the EGFR dimerization domain, which hinders dimerization with other HER family members.
Cetuximab (Erbitux®) uses
Preclinical studies have shown that cetuximab enhances the anti-tumour effects of chemotherapy (e.g. that of irinotecan in colorectal cancer) as well as radiotherapy (e.g. in squamous cell carcinoma of the head and neck) by inhibiting cell proliferation, angiogenesis and metastasis and by promoting apoptosis is used for the treatment of metastatic colorectal cancer, metastatic non-small cell lung cancer and head and neck cancer. Cetuximab also blocks growth factor-induced activation of the downstream mitogen-activated protein kinase, inhibiting cell proliferation. It has been also illustrated that cetuximab increases the receptor internalization which is another mechanism to silence the receptor. Cetuximab arrests cell cycle at G1 gap phase by upregulating antiproliferative p27 kip1 , which functions via complex formation with Cdk2, and downregulating proliferating cell nuclear antigen (PCNA).
Cetuximab also decreases angiogenic factors, inhibits tumor-cell invasion and metastasis via downregulation of matrix metalloproteinases (MMPs) and VEGF, and promotes apoptosis by upregulating apoptotic protein, Bax, with the help of other chemotherapeutic agents. Cetuximab has been widely shown to display synergistic effect with other agents and/or radiotherapy. Binding of antigen-binding fragment (Fab) of Cetuximab, which displays higher affinity comparing to ligands of EGFR, takes place via domain III of extracellular EGFR, preventing the receptor from conformational change to be dimerized and blocking EGFR signalling through inhibition of EGF and TGF-alpha-stimulated phosphorylation of the receptor.
Cetuximab (Erbitux®) and Epidermal Growth Factor Receptor
Epidermal growth factor receptor (EGFR) Epidermal growth factor receptor (EGFR; HER1; ErbB1) is a transmembrane tyrosine kinase encoded by Erb-B proto-oncogene, expressed in normal and malignant cells and stimulated by epidermal growth factor (EGF) or transforming growth factor-alpha (TGF-alpha) binding extracellular domain of the receptor, leading receptor to dimerize and activating intracellular kinase domain on each receptor, bringing about phosphorylation of tyrosine residues on each member of the receptor pair. Then, signalling complexes form in cytoplasm to activate gene transcription responding for such as cell proliferation. Termination of signalling occurs through internalization of receptor-ligand complex. Activation of EGFR results in perturbation of mitogen-activated protein kinase (MAPK), phosphatidylinositol 3-kinase, and AKT pathways triggering tumorigenic processes, such as increased proliferation, angiogenesis and metastasis, and prevents apoptosis.
Breast, lung, colon, prostate, kidney, bladder, head and neck, and ovary cancers have been associated to EGFR overexpression which causes early disease progression, poor survival, and resistance to chemotherapy in many epithelial malignancies. Epidermal growth factor receptor/human epidermal growth factor receptor 1 (EGFR/HER1) and its ligand, transforming growth factor alpha (TGF-alpha) were showed to involve in hepatocarcinogenesis. EGFR is overexpressed in hepatocellular carcinoma (HCC). To overcome the uncontrollable effect of EGFR triggering cancer development, monoclonal antibodies, such as Cetuximab, have been shown to be used as blockers in vitro and in vivo.
Cetuximab (Erbitux®) ELISA Kit Contents
| Size | Kit Contents |
| 1x12x8 | Microtiter Plate Break apart strips. Microtiter plate with 12 rows each of 8 wells coated with reactant. |
| 7 x 0.3 mL | Cetuximab Standards A-E (100x), High Level Control (100x), Low Level Control (100x) 300; 100; 30; 10; 0 microgram/mL |
| 1 x 50 mL | Assay Buffer |
| 1 x 12 mL | Horse radish peroxidase-Conjugate. Red coloured. Ready to use. Contains HRP-conjugate, stabilizer and preservatives. |
| 1 x 12 mL | TMB Substrate Solution |
| 1 x 12 mL | TMB Stop Solution |
| 1 x 50 mL | Wash Buffer concentrate (20x) |
| 2 x 1 | Adhesive Foil |
Cetuximab (Erbitux®) ELISA Protocol
| Steps | Protocol |
| 1 | Dilute each of the standards and samples (serum/plasma) at 1:100 using Assay Buffer as described in “Dilution of Standards and Samples (serum/plasma)” section of the technical manual. |
| 2 | Pipette 100µl of Assay Buffer non-exceptionally into each of the wells to be used. |
| 3 | Pipette 10 µL of each ready-to use Standards, High Level Control, Low Level Control and Diluted Samples into the respective wells of microtiter plate. |
| 4 | Cover the plate with adhesive foil. Incubate 30 min at room temperature (18- 25°C). |
| 5 | Remove adhesive foil. Discard incubation solution. Wash plate 3 times each with 300µL of diluted. Wash Buffer. Remove excess solution by tapping the inverted plate on a paper towel. |
| 6 | Pipette 100 µL of ready-to use HRP-Conjugate into each well. |
| 7 | Cover the plate with adhesive foil. Incubate 30 min at room temperature (18- 25°C). |
| 8 | Remove adhesive foil. Discard incubation solution. Wash plate 3 times each with 300 µL of diluted Wash Buffer. Remove excess solution by tapping the inverted plate on a paper towel. |
| 9 | Pipette 100 µL of TMB Substrate Solution into each well. |
| 10 | Incubate 10 min (without adhesive foil) at room temperature (18-25°C) in the dark |
| 11 | Stop the substrate reaction by adding 100 µL of Stop Solution into each well. Briefly mix contents by gently shaking the plate. Colour changes from blue to yellow. |
| 12 | Measure optical density with a photometer at 450/650 nm within 30 min after pipetting of the Stop Solution. |
Trademarks
Erbitux® CETUXIMAB is a trademark of ImClone LLC