Description
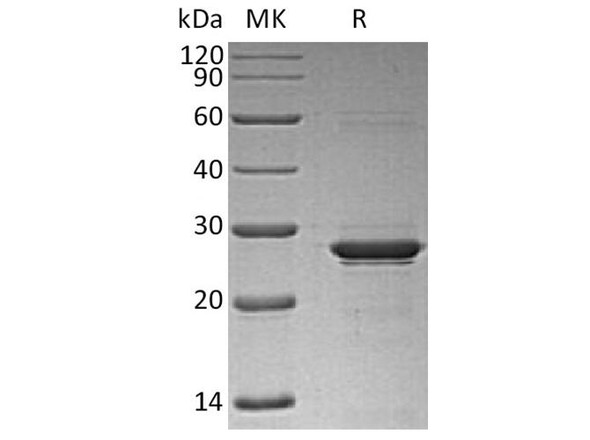
Recombinant Canine Oncostatin M Protein (KPRT0372)
| Product SKU | KPRT0372 |
| Product Type | Recombinant Protein |
| Quantity | 5 ug |
| Species | Canine |
| Target | Oncostatin M |
| Alias | OSM; |
| Source | Yeast |
| No. Amino Acids | 181 |
| Protein Sequence | SCSDKYPELL GQLQKQADFM QHTNTLLDLY IRSQGLDKNG LKEHCRERPG AFPSKDALQR LSRRVFLRTL DTTLGQVLLR LAALEQDIPK AQDLEMLSGV KLNIRGFKNN IHCMAQLLPG SSETTEPTPT SPGASPSPTP TLDTFQRRLE GCRFLHGYHR FMRSVGQVFR EWGKSLSRSR R (181) |
| MW | 20.6 kDa |
| Form | Lyophilized |
| Storage | -20°C |
| Shipping Conditions | Room temperature |