CIDE: The New Bioinformatics Engine Unmasking Secreted Proteins for Precision Immunotherapy
For decades, the battle against cancer has focused on the cellular 'soldiers' and the genetic 'blueprints' within them. Yet, a vast, invisible world of communication exists between these cells—a secret language of proteins released into the extracellular space. These secreted molecules often hold the key to why some patients respond to immunotherapy while others do not, but until now, mapping this complex 'secretome' has been an insurmountable challenge for modern oncology.
Introduction
The landscape of cancer treatment was forever changed by the advent of immunotherapy, yet the 'holy grail' of precision medicine remains elusive: the ability to predict with certainty which patient will benefit from which therapy. While genomic sequencing has provided a map of the tumor's internal mutations, it often fails to capture the dynamic interactions occurring in the tumor microenvironment. This is where the secretome—the collection of all proteins secreted by a cell—comes into play. These proteins act as messengers, modifiers, and sometimes, saboteurs of the immune response. Understanding this hidden layer of biological data is critical because the tumor secretome shapes the immune landscape during the relentless progression of the disease. By decoding these signals, researchers can identify new biomarkers and therapeutic targets that were previously invisible to standard diagnostic tools. The challenge, however, has always been the sheer volume and complexity of this data, requiring a new generation of computational tools to sift through the noise and find the signals that truly matter for patient survival.
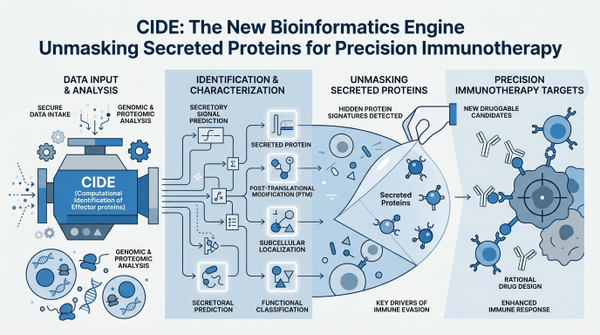
CIDE: The Computational Engine Decoding the Secretome
To bridge the gap between massive multi-omics datasets and clinical utility, researchers have developed a groundbreaking platform known as the Cancer Immunology Data Engine (CIDE). This isn't just another database; it is a sophisticated analytical engine designed to integrate thousands of patient profiles across dozens of tumor types. By synthesizing information from transcriptomics, proteomics, and clinical outcomes, CIDE identifies AOAH as immunotherapy potentiator, marking a significant leap forward in our ability to mine biological data for therapeutic gold. The engine works by systematically evaluating which genes, when expressed and secreted, correlate most strongly with positive responses to checkpoint inhibitors and other immunotherapies. This high-throughput approach allows scientists to move beyond single-gene studies and instead view the tumor microenvironment as a holistic, interconnected system. Interestingly, the engine's ability to prioritize secreted proteins is particularly valuable because these molecules are often more accessible for diagnostic testing and therapeutic targeting than intracellular proteins.
From Big Data to Biological Insight
The power of CIDE lies in its ability to turn abstract data into actionable biological hypotheses. By analyzing over 8,000 profiles, the engine can filter out the 'background noise' of the tumor and focus on the specific drivers of immune evasion or activation.
- Integration of 90+ omics datasets across 17 solid tumor types
- Prioritization of secreted proteins based on clinical outcome correlation
- Identification of novel metabolic regulators of the immune response
Unmasking AOAH: A Hidden Ally in the T-Cell Arsenal
One of the most compelling discoveries facilitated by the CIDE platform is the role of Acyloxyacyl Hydrolase (AOAH). While previously known for its role in detoxifying bacterial endotoxins, AOAH has emerged as a critical player in the success of cancer immunotherapy. The research reveals that AOAH isn't just a passive bystander; it actively reshapes the lipid environment of the tumor to favor immune activity. Specifically, AOAH works by depleting certain inhibitory lipids that would otherwise dampen the sensitivity of T-cell receptors. This metabolic 'cleanup' ensures that T-cells remain vigilant and capable of recognizing even weak tumor antigens. Beyond this, the presence of AOAH in the tumor microenvironment serves as a powerful predictor of how well a patient might respond to treatment. This finding is a perfect example of how 'dark data'—genes we've known about for years but didn't fully understand in a cancer context—can be brought into the light through advanced bioinformatics.
The Metabolic Sensitivity of T-Cells
The mechanism by which AOAH functions highlights a growing theme in oncology: the intersection of metabolism and immunology. By altering the lipid composition surrounding immune cells, AOAH directly impacts their functional state.
- Depletion of arachidonoyl phosphatidylcholines to boost TCR sensitivity
- Sensitization of T-cells to low-abundance tumor antigens
- Correlation between high AOAH expression and extended overall survival
The Secret Language of Macrophages and Immune Suppression
While AOAH acts as a potentiator, other secreted proteins can act as powerful brakes on the immune system. The complexity of these interactions is further revealed through single-cell analysis, which shows that the tumor microenvironment is a mosaic of competing signals. For instance, single-cell sequencing reveals immune response features that differ significantly between responders and non-responders, particularly in how macrophages are 'programmed' by the tumor. In some cases, secreted proteins like Clever-1 can actively suppress T-cell activity, creating a 'cold' tumor environment that is resistant to therapy. Recent studies have shown that secreted Clever-1 impacts immunotherapy efficacy by modulating the functional state of surrounding T-cells. Understanding these inhibitory signals is just as important as finding the activators, as it allows for the development of combination therapies that can simultaneously 'step on the gas' and 'release the brakes.'
Macrophages as Double Agents
Macrophages are often recruited by tumors to help suppress the immune response, but they can also be 'reprogrammed' to fight the cancer if the right signals are provided.
- Identification of SPP1+ macrophage populations associated with poor prognosis
- Role of secreted factors in driving macrophage polarization
- Potential for targeting secreted Clever-1 to 'heat up' cold tumors
Restoring the Balance: How Secreted PTEN Drives Immunity
The narrative of the secretome isn't just about what the tumor does to hide; it's also about how the body's natural defenses can be bolstered. Interestingly, even well-known tumor suppressors like PTEN have a 'secret' life outside the cell. While we typically think of PTEN as an intracellular regulator of cell growth, new evidence suggests that secreted PTEN drives macrophage antitumor immunity by binding to specific receptors like PLXDC2. This extracellular role for PTEN represents a paradigm shift in our understanding of tumor suppressors. It suggests that the loss of PTEN in a tumor doesn't just affect the cancer cell's internal growth pathways; it also strips the surrounding environment of a critical signal that would otherwise recruit and activate 'friendly' macrophages. By identifying these extracellular functions, researchers can explore new ways to deliver these protective signals back into the tumor microenvironment, potentially restoring the immune system's ability to recognize and destroy the cancer.
A New Frontier for Tumor Suppressors
The discovery of secreted PTEN's role opens up a new category of therapeutic opportunities where we treat the environment, not just the cell.
- PTEN-PLXDC2 interaction as a novel immune checkpoint
- Activation of pro-inflammatory pathways in tumor-associated macrophages
- Implications for PTEN-deficient tumors in immunotherapy resistance
The Future of Precision Immunotherapy
The emergence of tools like the Cancer Immunology Data Engine (CIDE) marks the beginning of a new era in precision oncology. By unmasking the secretome, we are moving beyond a two-dimensional view of cancer and entering a three-dimensional world of cellular conversation. The identification of proteins like AOAH and the extracellular roles of PTEN provide more than just new biomarkers; they offer a roadmap for the next generation of immunotherapies. As we continue to decode the secret language of the tumor microenvironment, the goal of truly personalized cancer treatment moves from a distant hope to a tangible reality. The secretome was once a hidden landscape, but thanks to the marriage of big data and biological insight, it is now becoming our most powerful weapon in the fight against cancer.
References
- Gong L, Luo J, et al. (2025). Cancer immunology data engine reveals secreted AOAH as a potential immunotherapy. Cell. 188(18):5062-5080.e32. PMID: 40730154
- Zhang C, et al. (2024). Secreted PTEN binds PLXDC2 on macrophages to drive antitumor immunity and tumor suppression. Dev Cell. 59(23):3072-3088.e8. PMID: 39197453
- Prince S, et al. (2025). Secreted Clever-1 modulates T cell responses and impacts cancer immunotherapy efficacy. Theranostics. 15(15):7501-7527. PMID: 40756372
- Yang J, et al. (2025). Tumor secretome shapes the immune landscape during cancer progression. J Exp Clin Cancer Res. 44(1):47. PMID: 39930476
- Yang Z, et al. (2024). Single-cell sequencing reveals immune features of treatment response to neoadjuvant immunochemotherapy in esophageal squamous cell carcinoma. Nat Commun. 15(1):8977. PMID: 39438438
Recent Posts
-
CIDE: The New Bioinformatics Engine Unmasking Secreted Proteins for Precision Immunotherapy
For decades, the battle against cancer has focused on the cellular 'soldiers' and the genetic 'blue …27th Mar 2026 -
The Bioelectronic Revolution: How Vagus Nerve Stimulation is Redefining Autoimmune Therapy in 2026
Imagine a future where chronic autoimmune diseases, once managed by powerful pharmaceuticals with c …25th Mar 2026 -
In Vivo CAR-T Engineering: The Dawn of Off-the-Shelf Immunotherapy
Imagine a future where cancer treatment isn't a grueling, months-long process involving cell extrac …25th Mar 2026