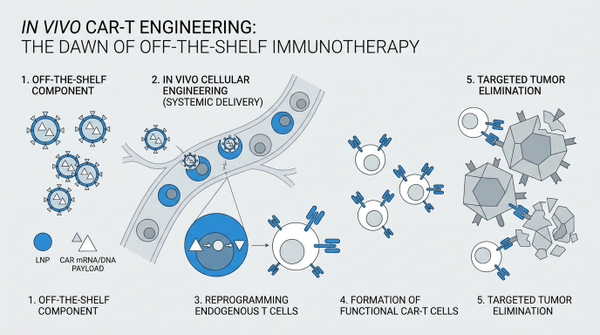
In Vivo CAR-T Engineering: The Dawn of Off-the-Shelf Immunotherapy
Imagine a future where cancer treatment isn't a grueling, months-long process involving cell extraction and reinfusion, but a simple, targeted injection that reprograms your body's own immune cells to fight disease from within. This isn't science fiction; it's the rapidly emerging reality of in vivo CAR-T engineering, a revolutionary approach poised to transform immunotherapy and bring 'off-the-shelf' treatments closer than ever before.
Introduction
Chimeric Antigen Receptor (CAR) T-cell therapy has already revolutionized the treatment landscape for certain blood cancers, offering a powerful new weapon against previously intractable diseases. However, the traditional 'ex vivo' manufacturing process is complex, costly, and time-consuming. It involves extracting a patient's T cells, genetically modifying them in a lab to express CARs that recognize cancer cells, expanding these modified cells, and then reinfusing them back into the patient. This intricate process limits accessibility and can delay critical treatment for patients in urgent need. The dawn of in vivo CAR-T engineering promises to overcome these significant hurdles by enabling the direct reprogramming of T cells within the body, bypassing the need for external manipulation and offering a more streamlined, accessible, and potentially safer therapeutic strategy. This innovative approach represents a paradigm shift, moving us closer to truly 'off-the-shelf' immunotherapies that could be readily available to a broader patient population, impacting not only oncology but also autoimmune diseases.
From Lab Bench to Living System: The Shift to In Vivo Reprogramming
The conventional CAR-T cell therapy, while remarkably effective, is burdened by its elaborate manufacturing pipeline. The logistical challenges, high costs, and the time required for ex vivo cell processing often create bottlenecks, making it inaccessible for many patients globally. This is where the concept of in vivo CAR-T engineering emerges as a game-changer. Instead of modifying T cells outside the body, researchers are now developing ingenious methods to deliver genetic instructions directly to T cells within the patient's bloodstream or tissues. This direct approach not only simplifies the therapeutic process but also holds the potential to reduce treatment costs and accelerate patient access to life-saving immunotherapies. The goal is to create a more efficient and scalable solution, transforming the complex 'made-to-order' model into a more readily available 'off-the-shelf' treatment strategy. Direct body production overcomes high manufacturing costs, addressing a critical limitation of current CAR-T therapies. Furthermore, nanoparticle-formulated nucleic acids bypass ex vivo manufacturing, paving the way for a new era of cellular therapies.
The Architects of Change: Viral Vectors and Lipid Nanoparticles
The success of in vivo CAR-T engineering hinges on the efficient and safe delivery of genetic material to T cells. Scientists are primarily exploring two sophisticated delivery systems: viral vectors and lipid nanoparticles (LNPs). Viral vectors, particularly lentiviral vectors, have been engineered to precisely target T cells and deliver the CAR gene. Recent advancements have focused on creating fourth-generation lentiviral vectors enable efficient T-cell generation, demonstrating improved efficiency and safety profiles. These vectors can be designed to specifically transduce T cells, minimizing off-target effects. Concurrently, lipid nanoparticles have emerged as a highly promising non-viral alternative. These tiny lipid spheres encapsulate messenger RNA (mRNA) encoding the CAR, which is then delivered to T cells. Extrahepatic lipid nanoparticles enable transient CAR expression, offering a transient yet potent reprogramming of T cells. This transient expression can be advantageous for managing potential toxicities and providing a more controllable therapeutic window. Moreover, novel viral fusogens enable targeted genetic medicine delivery, further enhancing the precision of in vivo engineering.
Early Triumphs and Broadening Horizons
Preclinical studies are already showcasing the immense potential of in vivo CAR-T engineering. Researchers have successfully demonstrated that targeted lipid nanoparticles reprogram T cells in vivo, leading to the generation of functional CAR T cells that can effectively target and eliminate tumors in animal models. These studies are not only validating the feasibility of the approach but also revealing its potential applicability beyond oncology. Intriguingly, in vivo CAR-T engineering is also being explored for autoimmune diseases, where reprogramming T cells could offer novel therapeutic avenues. The ability to generate CAR T cells directly within the body eliminating ex vivo processing improves therapy logistics, making it a more practical solution for a wider range of conditions. The initial successes in preclinical models are paving the way for clinical trials, bringing us closer to realizing the full therapeutic promise of this groundbreaking technology. The development of lentiviral platform enables dose-dependent in vivo transduction, further solidifying the foundation for clinical translation.
The Future is In Vivo: A New Era for Immunotherapy
The journey from traditional ex vivo CAR-T therapy to in vivo CAR-T engineering marks a pivotal moment in the history of immunotherapy. By overcoming the logistical and financial barriers of current treatments, this innovative approach promises to democratize access to advanced cellular therapies. While challenges remain in optimizing delivery systems, ensuring long-term safety, and refining targeting specificity, the rapid pace of research suggests a future where 'off-the-shelf' CAR-T treatments are not just a possibility, but a widespread reality. This shift will not only benefit cancer patients but also open new doors for treating a spectrum of other diseases, heralding a new era of precision medicine where the body's own defenses are powerfully and precisely orchestrated from within.
References
- Hunter TL, Bao Y, Zhang Y, et al. (2025). In vivo CAR T cell generation to treat cancer and autoimmune disease.. Science. 388(6753):1311-1317. PMID: 40536974
- Billingsley MM, Gong N, Mukalel AJ, et al. (2024). In Vivo mRNA CAR T Cell Engineering via Targeted Ionizable Lipid Nanoparticles with Extrahepatic Tropism.. Small. 20(11):e2304378. PMID: 38072809
- Short L, Holt RA, Cullis PR, Evgin L. (2024). Direct in vivo CAR T cell engineering.. Trends Pharmacol Sci. 45(5):406-418. PMID: 38614815
- Bui TA, Mei H, Sang R, et al. (2024). Advancements and challenges in developing in vivo CAR T cell therapies for cancer treatment.. EBioMedicine. 106:105266. PMID: 39094262
- Bot A, Scharenberg A, Friedman K, et al. (2026). In vivo chimeric antigen receptor (CAR)-T cell therapy.. Nat Rev Drug Discov. 25(2):116-137. PMID: 41028170
- Michels KR, Sheih A, Hernandez SA, et al. (2023). Preclinical proof of concept for VivoVec, a lentiviral-based platform for in vivo CAR T-cell engineering.. J Immunother Cancer. 11(3):e006292. PMID: 36918221
- Coradin T, Keating AL, Barnard AR, et al. (2025). Efficient in vivo generation of CAR T cells using a retargeted fourth-generation lentiviral vector.. Mol Ther. 33(10):4953-4967. PMID: 40676833
- Andorko JI, Russell RM, Schnepp BC, et al. (2025). Targeted in vivo delivery of genetic medicines utilizing an engineered lentiviral vector platform results in CAR T and NK cell generation.. Mol Ther. 33(10):4937-4952. PMID: 40581818
Recent Posts
-
The Bioelectronic Revolution: How Vagus Nerve Stimulation is Redefining Autoimmune Therapy in 2026
Imagine a future where chronic autoimmune diseases, once managed by powerful pharmaceuticals with c …25th Mar 2026 -
In Vivo CAR-T Engineering: The Dawn of Off-the-Shelf Immunotherapy
Imagine a future where cancer treatment isn't a grueling, months-long process involving cell extrac …25th Mar 2026 -
Immune Cell 'Bloodhounds': Engineering Metabolite-Sensing Receptors to Infiltrate Solid Tumors
Imagine an elite squad of soldiers sent to neutralize a hidden fortress, only to find themselves wa …25th Mar 2026