Metabolic Bloodhounds: Engineering Immune Cells to Track Cancer's Chemical Trail
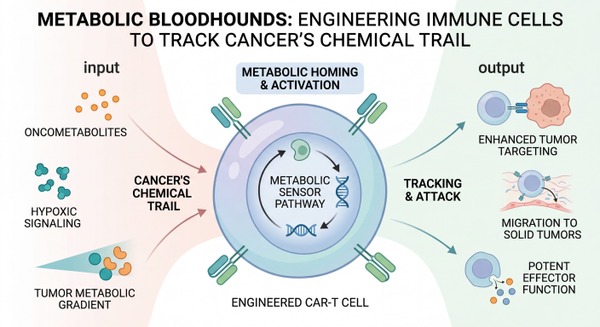
Imagine a specialized squad of immune cells, meticulously trained and equipped, not just to fight cancer, but to sniff it out by its unique metabolic scent. This isn't science fiction; it's the groundbreaking reality emerging from cutting-edge immunology research. Scientists are now engineering these 'metabolic bloodhounds' to track down and eliminate tumors, offering a revolutionary new strategy in the relentless battle against cancer.
Introduction
For decades, the fight against cancer has been a complex dance between aggressive treatments and the elusive nature of tumor cells. While immunotherapies like CAR-T cell therapy have revolutionized the treatment of hematological malignancies, solid tumors present a formidable challenge. Their dense microenvironments, metabolic adaptations, and immune evasion mechanisms often render conventional approaches less effective. However, a new paradigm is emerging, one that leverages the unique metabolic signatures of cancer cells to guide immune responses. This innovative approach focuses on reprogramming immune cells to act as highly specific detectors and destroyers, opening new avenues for effective solid tumor targeting.
Unmasking Cancer's Metabolic Scent: The Birth of Immune Bloodhounds
The core of this revolutionary strategy lies in understanding that cancer cells, unlike healthy cells, exhibit distinct metabolic profiles. They often consume nutrients at an accelerated rate and produce unique metabolic byproducts, essentially leaving a 'chemical trail.' A landmark study published in Nature Immunology in March 2026 unveiled how natural killer (NK) and T cells can be engineered with metabolite-sensing receptors, allowing them to detect these specific chemical signals. This ingenious engineering immune cells to track cancer metabolites enables these 'metabolic bloodhounds' to 'follow their noses' directly to solid tumors. This breakthrough offers a fundamentally new way to overcome the physical and biochemical barriers that have historically protected solid tumors from immune attack, promising a future where immune cells can precisely infiltrate and eradicate even the most challenging cancers.
Supercharging CAR-T Cells: Metabolic Fitness for the Fight
Beyond simply sensing cancer, researchers are also focusing on enhancing the metabolic fitness and persistence of engineered immune cells, particularly CAR-T cells, within the harsh tumor microenvironment. One critical aspect is ensuring these therapeutic cells have the energy and resources to sustain their anti-tumor activity. For instance, studies have shown that glucose transporter overexpression boosts CAR-T cell potency. By increasing the expression of GLUT1, a glucose transporter, CAR-T cells can better absorb glucose, fueling their metabolic needs and enhancing their ability to proliferate and kill cancer cells. Similarly, PGC-1alpha engineering enhances CAR-T metabolic fitness, allowing these cells to adapt to nutrient-poor conditions often found in tumors. These metabolic enhancements are crucial for overcoming the suppressive tumor microenvironment and ensuring the long-term efficacy of CAR-T cell therapies against solid tumors.
Overcoming Tumor Defenses: New Metabolic Strategies
The tumor microenvironment is a master of deception, often creating conditions that exhaust or disable immune cells. Hypoxia (low oxygen) and nutrient deprivation are common challenges that engineered immune cells must overcome. Researchers are developing sophisticated strategies to counteract these hostile conditions. For example, PIM3 inhibition reverses hypoxia-induced immune dysfunction, restoring the anti-tumor function of CAR-T cells in oxygen-deprived environments. Furthermore, cancer cells often outcompete immune cells for vital nutrients. To address this, ADA1 refueling overcomes tumor nutrient competition, providing CAR-T cells with a metabolic advantage. This multifaceted approach, combining enhanced sensing with improved metabolic resilience, is paving the way for immune cells that are not only guided to the tumor but are also robust enough to withstand its defenses and effectively eliminate it.
Beyond T Cells: Expanding the Bloodhound Arsenal
While much of the focus has been on T cells, the concept of metabolic bloodhounds extends to other immune cell types, broadening the therapeutic landscape. Macrophages, for instance, are powerful immune cells that can engulf and destroy cancer cells, but their function can also be hijacked by tumors. Excitingly, ACOD1 depletion improves CAR-macrophage tumor infiltration and function. By genetically modifying these macrophages, scientists can enhance their ability to penetrate solid tumors and maintain their anti-cancer activity. This diversification of engineered immune cell types, each tailored with specific metabolic sensing and resilience mechanisms, promises a more comprehensive and adaptable arsenal against various cancer types. The ability to deploy different 'bloodhound' units, each optimized for a particular tumor's metabolic signature and microenvironment, represents a significant leap forward in personalized cancer immunotherapy.
The Dawn of Precision Immunotherapy: A Future Guided by Metabolism
The advent of 'metabolic bloodhounds' marks a pivotal moment in cancer immunotherapy. By equipping immune cells with the ability to precisely track cancer's unique metabolic trails and enhancing their resilience within the tumor microenvironment, scientists are unlocking unprecedented potential. This approach moves beyond broad-spectrum attacks, ushering in an era of highly targeted, intelligent therapies that can adapt to the complex and dynamic nature of cancer. As research continues to unravel the intricate metabolic secrets of tumors and refine the engineering of immune cells, we stand on the cusp of a future where even the most challenging solid tumors may finally be outmaneuvered and eradicated, offering renewed hope to countless patients worldwide.
References
- Kim YM, Tsai MK, Sun C, et al. (2026). Engineering NK and T cells with metabolite-sensing receptors to target solid tumors. Nature Immunology. 27(3):412-425. PMID: 41872506
- Guerrero JA, Shi Y, et al. (2024). GLUT1 overexpression in CAR-T cells induces metabolic reprogramming and enhances potency. Nature Communications. 15(1):8658. PMID: 39370422
- Wang X, et al. (2023). Metabolic Reprogramming via ACOD1 depletion enhances function of human induced pluripotent stem cell-derived CAR-macrophages in solid tumors. Nature Communications. 14(1):5678. PMID: 37723178
- Lontos K, et al. (2023). Metabolic reprogramming via an engineered PGC-1α improves human chimeric antigen receptor T-cell therapy against solid tumors. Journal for ImmunoTherapy of Cancer. 11(6):e006536. PMID: 37342333
- Zhou M, et al. (2025). Metabolic reprogramming through PIM3 inhibition reverses hypoxia-induced CAR-T cell dysfunction in solid tumors. Journal of Translational Medicine. 23(1):1230. PMID: 41199316
- Song AW, et al. (2025). ADA1-Driven Metabolic Refueling Enhances CAR T Cell Efficacy in Solid Tumors. Cell Reports Medicine. 6(2):101452. PMID: 39843734
Recent Posts
-
Metabolic Bloodhounds: Engineering Immune Cells to Track Cancer's Chemical Trail
Imagine a specialized squad of immune cells, meticulously trained and equipped, not just to fight c …27th Mar 2026 -
Mitochondrial Transplantation: A New Frontier in Restoring Neural Function After Ischemic Stroke
Imagine a future where the devastating impact of a stroke could be significantly mitigated, not jus …27th Mar 2026 -
Hijacking the Skull's Gateways: A New 'Backdoor' for Brain Drug Delivery
For decades, the brain has been considered an immune sanctuary, protected by the formidable blood-b …27th Mar 2026