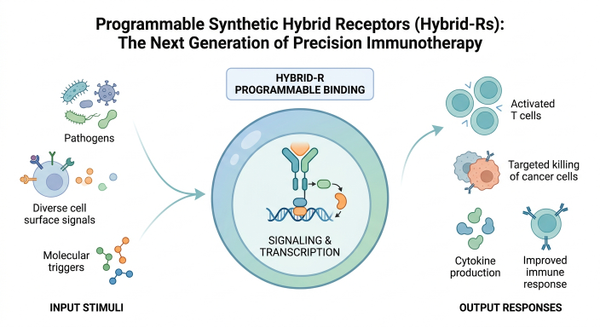
Programmable Synthetic Hybrid Receptors (Hybrid-Rs): The Next Generation of Precision Immunotherapy
Imagine a world where cancer treatment is no longer a blunt instrument but a precision-guided surgical strike, executed by the body's own immune system. For years, we have dreamed of 'smart' cells that can navigate the complex terrain of a tumor, distinguish friend from foe with absolute certainty, and adapt their behavior in real-time. Today, that dream is rapidly becoming a reality through the emergence of Programmable Synthetic Hybrid Receptors, or Hybrid-Rs, which are redefining the very boundaries of immunotherapy.
Introduction
The landscape of modern oncology has been fundamentally altered by the advent of Chimeric Antigen Receptor (CAR) T-cell therapy. By genetically engineering a patient's own T cells to express synthetic receptors, scientists have created a living drug capable of seeking out and destroying malignant cells. This approach has seen remarkable success in treating liquid biopsies like B-cell leukemias and lymphomas. However, the transition to solid tumors has proven far more challenging. Tumors are not just masses of rogue cells; they are complex, hostile ecosystems that employ a variety of tactics to evade, suppress, and even kill the immune cells sent to destroy them. Traditional CAR-T cells often struggle with 'off-tumor' toxicity, where they attack healthy tissues expressing low levels of the target antigen, or they succumb to the immunosuppressive signals of the tumor microenvironment. As we look toward the future, it is clear that synthetic receptors revolutionize modern clinical oncology by providing the modularity and control necessary to overcome these hurdles. The next generation of this technology, the Hybrid-R, represents a leap forward in our ability to program immune cells with the logic and precision of a computer.
The Architecture of Intelligence: How Hybrid-Rs Merge CAR and SNIPR Logic
At its core, the Hybrid-R is a masterpiece of biological engineering. It doesn't just copy existing designs; it integrates the best features of two distinct receptor classes: the Chimeric Antigen Receptor (CAR) and the Synthetic Notch (synNotch) receptor. While a standard CAR provides the 'go' signal for killing, it often lacks the nuance required for complex decision-making. In contrast, synNotch receptors allow for 'if-then' logic, but they don't directly trigger the T cell's killing machinery. The Hybrid-R bridges this gap by incorporating a SNIPR-like intramembrane proteolysis mechanism directly into a signaling-competent receptor framework. This means that Hybrid-Rs enable precise context-dependent control over T-cell activity. When the receptor binds to its target, it doesn't just activate the cell; it can also trigger the release of a transcriptional activator that travels to the nucleus to turn on specific genes. This allows scientists to program T cells that only become fully active when they encounter a specific combination of signals, effectively creating a biological 'logic gate' that ensures the therapy only hits its intended target.
Breaking the Siege: Overcoming the Hostile Tumor Microenvironment
One of the greatest obstacles in treating solid tumors is the 'hostile neighborhood' they create. Tumors often surround themselves with a shield of immunosuppressive molecules and 'death ligands' like FasL, which can trigger apoptosis in incoming T cells. To survive this onslaught, engineered cells need more than just a target; they need a shield and a booster. Recent research has shown that we can engineer 'chimeric' versions of these death receptors to turn a negative signal into a positive one. For instance, Fas-TNFR chimeras overcome hostile tumor microenvironments by redirecting the Fas-mediated 'kill' signal into a pro-survival and activation signal. Beyond just surviving, these cells must also expand and persist within the tumor. To address this, scientists have developed 'Turbodomains'—synthetic cytokine receptor fragments that provide a constant, low-level growth signal. These Turbodomains enhance expansion without systemic toxicity, allowing the T cells to maintain their numbers and antitumor activity without the dangerous side effects often associated with systemic cytokine administration.
The Precision of MHC-Independent Recognition
A significant limitation of natural T-cell recognition is its dependence on the Major Histocompatibility Complex (MHC), which tumors frequently downregulate to hide from the immune system. Synthetic receptors bypass this by using antibody-derived domains to recognize surface proteins directly. However, simply bypassing MHC isn't enough; the recognition must be incredibly sensitive and specific to avoid 'on-target, off-tumor' effects. This is where rational protein engineering comes into play. By fine-tuning the affinity and structure of these receptors, researchers have found that MHC-independent receptors re-engage precise immune responses that were previously impossible. This precision is further enhanced by the development of bispecific receptors, which require the presence of two different antigens on the same cell to trigger a full response. This 'dual-key' system is a game-changer because bispecific receptors prevent antigen-negative tumor relapse, a common failure mode where tumors simply stop expressing the single target antigen to escape the therapy.
Key Advantages of the Hybrid-R Platform
The Hybrid-R platform offers several distinct advantages over traditional CAR-T designs, making it a versatile tool for the next generation of immunotherapy.
- Modular Design: Easily swap extracellular domains to target different cancer types.
- Programmable Payloads: Trigger the local production of cytokines or antibodies only at the tumor site.
- Enhanced Safety: Logic-gated activation minimizes the risk of attacking healthy tissue.
- Improved Persistence: Integrated survival signals help T cells endure the tumor microenvironment.
Conclusion: The Dawn of Programmable Medicine
The journey from basic immunology to the engineering of Hybrid-Rs represents one of the most exciting chapters in modern medicine. We are moving away from a 'one-size-fits-all' approach and toward a future where every patient's therapy can be custom-programmed to navigate their specific tumor landscape. While challenges remain—particularly in scaling these complex therapies and ensuring long-term safety—the progress made in just the last few years is staggering. Hybrid-Rs are not just a new type of receptor; they are a new way of thinking about disease. By treating the immune system as a programmable platform, we are unlocking the potential to treat not just cancer, but a wide range of autoimmune and infectious diseases. The era of precision, programmable immunotherapy has arrived, and the Hybrid-R is leading the charge.
A New Frontier in Cellular Engineering
As we reflect on the rapid evolution of synthetic biology, the Hybrid-R stands as a testament to human ingenuity. It represents the convergence of computing logic and biological function, offering a level of control that was once the stuff of science fiction. By integrating modular signaling, survival boosters, and multi-antigen logic, we are finally equipping our immune 'soldiers' with the intelligence they need to win the war against solid tumors. The road ahead will require continued collaboration between clinicians, engineers, and biologists, but the destination—a world where cancer is a manageable, if not curable, condition—is closer than ever before.
References
- Foisey MG, Garcia J, Li X, Yang X, Hilburger C, et al. (2026). Synthetic Hybrid Receptors for Safer and Programmable T Cell Therapy. bioRxiv. Preprint:10.64898/2026.01.22.701150v1. PMID: 41648153
- Simon S, Bugos G, Prins R, Rajan A, Palani A, et al. (2025). Design of sensitive monospecific and bispecific synthetic chimeric T cell receptors for cancer therapy. Nat Cancer. 6(4):647-665. PMID: 40097658
- Chang JF, Landmann JH, Chang TC, Selli ME, Tenzin Y, et al. (2024). Rational Protein Engineering to Enhance MHC-Independent T-cell Receptors. Cancer Discov. 14(11):2109-2121. PMID: 38980802
- McKenzie C, El-Kholy M, Parekh F, Robson M, Lamb K, et al. (2023). Novel Fas-TNFR chimeras that prevent Fas ligand-mediated kill and signal synergistically to enhance CAR T cell efficacy. Mol Ther Nucleic Acids. 32:603-621. PMID: 37200859
- Lin RJ, Sutton J, Bentley T, Vargas-Inchaustegui DA, Nguyen D, et al. (2023). Constitutive Turbodomains enhance expansion and antitumor activity of allogeneic BCMA CAR T cells in preclinical models. Sci Adv. 9(31):eadg8694. PMID: 37540748
- Kamli H, Khan NU. (2025). Revolutionising cancer intervention: the repercussions of CAR-T cell therapy on modern oncology practices. Med Oncol. 42(7):228. PMID: 40448746
31st Mar 2026
Recent Posts
-
Programmable Synthetic Hybrid Receptors (Hybrid-Rs): The Next Generation of Precision Immunotherapy
Imagine a world where cancer treatment is no longer a blunt instrument but a precision-guided surgi …31st Mar 2026 -
The Neural Shield: How Cancer-Induced Nerve Injury Drives Immunotherapy Resistance
For years, our understanding of cancer focused primarily on the tumor cells themselves and the immu …29th Mar 2026 -
Metabolic Bloodhounds: Engineering Immune Cells to Track Cancer's Chemical Trail
Imagine a specialized squad of immune cells, meticulously trained and equipped, not just to fight c …27th Mar 2026