Hijacking the Skull's Gateways: A New 'Backdoor' for Brain Drug Delivery
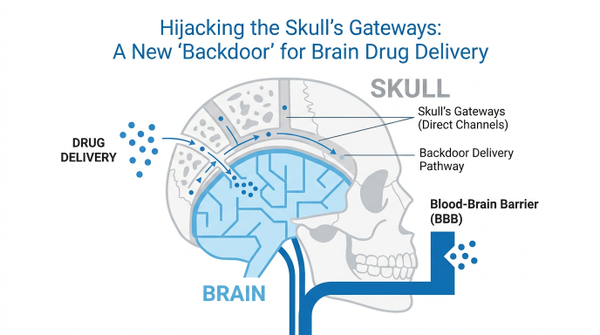
For decades, the brain has been considered an immune sanctuary, protected by the formidable blood-brain barrier. This intricate defense system, while vital for safeguarding our most complex organ, has simultaneously posed an immense challenge for delivering therapeutic drugs to treat neurological diseases. But what if there was a hidden "backdoor," a previously overlooked pathway offering a direct route into the brain? Recent groundbreaking research suggests such a gateway exists, nestled within the very bones of our skull.
Introduction
The concept of the brain as an "immune-privileged" site has long dominated neuroscience, largely due to the blood-brain barrier (BBB) – a highly selective semipermeable border that separates the circulating blood from the brain and extracellular fluid in the central nervous system (CNS). This barrier effectively blocks most large molecules and many small molecules, including potential therapeutic agents, from reaching brain tissue. Consequently, developing effective treatments for conditions like Alzheimer's, Parkinson's, and brain tumors has been a monumental task. However, a paradigm shift is underway. Emerging discoveries are revealing a complex network of channels within the skull bone marrow that act as direct conduits to the brain, fundamentally altering our understanding of CNS immunity and drug delivery. This newfound understanding promises to revolutionize how we approach neurological disorders, offering novel avenues for therapeutic intervention. Beyond its protective role, the skull is emerging as a dynamic interface that actively communicates with the brain, providing a unique vantage point for both diagnosis and treatment. By understanding how these channels function, we can begin to design therapies that 'hitchhike' on natural biological pathways, potentially bypassing the BBB altogether and delivering medicine exactly where it is needed most.
Unveiling the Skull's Secret Passageways: A Direct Route to the Brain
The skull, far from being a mere protective casing, is now recognized as a dynamic hub for immune surveillance and a potential conduit for therapeutic delivery. Recent studies have illuminated the intricate network of microscale ossified vascular channels that directly connect the skull bone marrow to the brain's meninges and even the brain parenchyma itself. These skull channels serve as immune gateways, challenging the long-held belief that the brain is entirely isolated from the peripheral immune system. Researchers have meticulously characterized these channels in both humans and mice, revealing their unique structural and cellular details. These findings indicate that the skull bone marrow is not just a site for hematopoiesis but an active participant in CNS immunity, capable of mobilizing immune cells and molecules directly to the brain. Interestingly, these channels appear to be more than just simple pipes; they are sophisticated biological interfaces that regulate the flow of information and materials. The implications are profound, suggesting that the skull acts as a critical interface, mediating communication between the peripheral immune system and the central nervous system. This redefines the landscape of CNS immune surveillance and opens up exciting possibilities for bypassing the traditional barriers to brain drug delivery. By mapping these pathways, scientists are discovering that the brain's 'isolation' was perhaps an illusion created by our limited imaging capabilities, and that the skull is actually a vibrant part of the brain's ecosystem.
The Anatomy of a Biological Backdoor
To understand how these gateways work, we must look at the microscopic architecture of the calvaria. These channels are not random gaps but highly organized structures that facilitate the rapid transit of myeloid cells from the marrow to the meninges during times of stress or injury. This direct line of communication allows the brain to recruit 'first responders' without waiting for them to travel through the general circulation, where they might be diluted or delayed.
- Direct vascular connections between marrow and dura mater
- Preferential recruitment of skull-derived neutrophils during inflammation
- Short-circuiting the systemic circulation for rapid immune response
Molecular Signatures and Inflammatory Responses: The Skull's Role in Neurological Health
Beyond the anatomical revelation, scientists have delved into the molecular intricacies of the skull bone marrow, uncovering distinct profiles that underscore its unique role in neurological health and disease. Research has shown that the mouse skull, in particular, exhibits a highly distinct transcriptomic profile compared to other bones, characterized by a late-stage neutrophil phenotype. In humans, proteomic analysis further confirms that skull marrow possesses a unique molecular signature, with differentially expressed neutrophil-related pathways and a specific synaptic protein signature. Crucially, skull marrow profiles reflect brain inflammation, suggesting that the skull bone marrow is not merely a passive reservoir but an active participant in the brain's immune responses. This molecular 'mirroring' means that what happens in the brain is often reflected in the skull's marrow, providing a potential non-invasive window into the brain's health. Furthermore, imaging studies have demonstrated that the skull bone marrow reflects inflammatory brain responses, exhibiting disease-specific spatial distributions in patients with various neurological disorders. This highlights the potential of the skull as a diagnostic and monitoring site for brain diseases, offering a window into the brain's inflammatory state without the need for invasive brain biopsies. Interestingly, the skull marrow seems to 'remember' past inflammatory events, potentially serving as a biological record of the brain's immunological history. This could lead to new ways of tracking disease progression in chronic conditions like multiple sclerosis or Alzheimer's disease, where understanding the timing and location of inflammation is key to effective treatment.
A Two-Way Street: Cellular and Solute Exchange Across the Meningeal Border
The traditional view of the arachnoid membrane as a strict barrier preventing direct passage of cells and large molecules between the cerebrospinal fluid (CSF) and bone marrow is being re-evaluated. A systematic review of recent literature provides compelling evidence for a potential two-way passage across the meningeal border. This review identified 16 studies demonstrating communication between the brain, meninges, and skull bone marrow, with cells like B and T cells, neutrophils, bacteria, and various drug compounds passing through. This exchange is critical for understanding CNS immunity and opens new avenues for drug delivery. The dynamic interplay between CSF transport and the brain-meningeal border also significantly impacts neurological health and disease, as CSF exchange impacts neurological disease progression. These findings collectively redefine our understanding of brain-immune interactions and highlight the skull's role in maintaining CNS homeostasis and responding to pathology. Beyond this, the discovery of these bidirectional pathways suggests that the brain might actually 'export' waste products or signaling molecules directly to the skull marrow for processing or systemic signaling. This could mean that the skull is part of a larger 'glymphatic-lymphatic' network that helps keep the brain clean and healthy. If we can learn to control this flow, we might be able to enhance the brain's natural waste-clearance mechanisms or deliver drugs that use these same exit routes as entry points, effectively 'swimming upstream' into the brain.
Redefining CNS Immune Surveillance: The Skull's Unique Contribution
The discovery of skull meninges channels (SMCs) has fundamentally reshaped our understanding of central nervous system (CNS) immunity. These channels, along with the skull bone marrow, are now recognized as critical components in redefining CNS surveillance via skull channels. The skull bone marrow-meninges border possesses unique aspects that enable immune cell and molecule trafficking, making it a distinct interface for brain-immune communication. As researchers have noted, these unique border features enable molecule trafficking, which is essential for the brain's ability to sense and respond to peripheral threats. This newfound understanding moves beyond the traditional concept of immune privilege, emphasizing a more interactive and dynamic relationship between the CNS and the peripheral immune system. The skull's contribution extends to both maintaining health and responding to various CNS diseases, suggesting that targeting these pathways could offer novel therapeutic strategies. For instance, by modulating the activity of immune cells within the skull marrow, we might be able to dampen neuroinflammation in the brain without the side effects associated with systemic immunosuppression. This localized approach to neuroimmunology could be a game-changer for patients with autoimmune or neurodegenerative conditions. Furthermore, the realization that the skull is an active immunological organ means we need to reconsider how we treat head injuries or perform cranial surgeries, as these interventions could inadvertently disrupt the brain's primary line of defense and communication.
The Future of 'Transcranial' Medicine
As we look toward the future, the possibility of 'transcranial' drug delivery—where medicine is applied to or injected into the skull bone to reach the brain—is becoming increasingly plausible. This would avoid the risks of direct brain injections and the inefficiencies of systemic delivery. Imagine a world where a simple patch on the scalp could deliver life-saving treatments for stroke or epilepsy, using the skull's own gateways to reach the target tissue.
- Development of bone-seeking nanoparticles for targeted delivery
- Non-invasive monitoring of brain health via skull-based sensors
- Localized immunotherapy for brain tumors using skull marrow niches
The Skull's Hidden Potential: A New Era for Brain Therapeutics
The revelation of the skull's intricate channels and its active role in brain immunity marks a pivotal moment in neuroscience. This "backdoor" for brain drug delivery, bypassing the formidable blood-brain barrier, offers unprecedented opportunities for treating a wide array of neurological disorders. From targeted drug delivery to modulating neuroinflammatory responses, the skull's gateways present a promising frontier. Further research into these pathways will undoubtedly unlock new therapeutic strategies, bringing us closer to effective treatments for conditions that have long eluded conventional approaches. The skull, once considered a passive protector, is now emerging as a key player in the future of brain health. As we continue to map these secret passageways, we are not just finding new ways to deliver drugs; we are discovering a more integrated, dynamic, and accessible brain than we ever imagined. The era of the 'impenetrable' brain is ending, and a new era of skull-mediated therapeutics is just beginning.
References
- Kolabas ZI, Kuemmerle LB, et al. (2023). Distinct molecular profiles of skull bone marrow in health and neurological disorders. Cell. 186(17):3706-3725.e29. PMID: 37562402
- Mazzitelli JA, Pulous FE, et al. (2023). Skull bone marrow channels as immune gateways to the central nervous system. Nat Neurosci. 26(12):2052-2062. PMID: 37996526
- Liu L, Zhang X, et al. (2025). Skull bone marrow and skull meninges channels: redefining the landscape of central nervous system immune surveillance. Cell Death Dis. 16(1):53. PMID: 39875352
- Therkelsen HE, Enger R, et al. (2025). Evidence for cellular and solute passage between the brain and skull bone marrow across meninges: A systematic review. J Cereb Blood Flow Metab. 45(4):581-599. PMID: 39862438
- Pinho-Correia LM, McCullough SJC, et al. (2025). CSF transport at the brain-meningeal border: effects on neurological health and disease. Lancet Neurol. 24(6):535-547. PMID: 40409317
- Gadani SP, Calabresi PA. (2023). The calvaria stands alone: Unique aspects of the skull bone marrow-meninges border. Cell. 186(17):3524-3526. PMID: 37595561
Recent Posts
-
Mitochondrial Transplantation: A New Frontier in Restoring Neural Function After Ischemic Stroke
Imagine a future where the devastating impact of a stroke could be significantly mitigated, not jus …27th Mar 2026 -
Hijacking the Skull's Gateways: A New 'Backdoor' for Brain Drug Delivery
For decades, the brain has been considered an immune sanctuary, protected by the formidable blood-b …27th Mar 2026 -
CIDE: The New Bioinformatics Engine Unmasking Secreted Proteins for Precision Immunotherapy
For decades, the battle against cancer has focused on the cellular 'soldiers' and the genetic 'blue …27th Mar 2026