Mitochondrial Transplantation: A New Frontier in Restoring Neural Function After Ischemic Stroke
Imagine a future where the devastating impact of a stroke could be significantly mitigated, not just by managing symptoms, but by directly repairing the cellular machinery damaged during the event. This isn't science fiction; it's the rapidly evolving reality of mitochondrial transplantation, a groundbreaking approach poised to redefine how we restore neural function after ischemic stroke.
Introduction
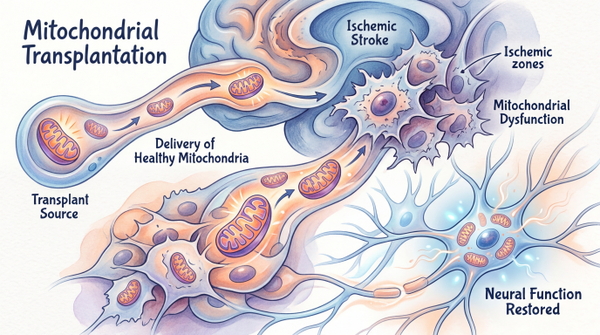
Ischemic stroke, caused by a blockage in blood flow to the brain, deprives neurons of vital oxygen and nutrients, leading to rapid cellular damage and death. The aftermath can leave individuals with profound and lasting neurological deficits. For decades, treatments have focused on restoring blood flow and protecting neurons from immediate harm. However, a new frontier in neuroscience is emerging, one that targets the very powerhouses of our cells: mitochondria. These tiny organelles are crucial for energy production, and their dysfunction is a hallmark of ischemic injury. By transplanting healthy mitochondria into damaged brain tissue, scientists aim to revitalize compromised cells, enhance their survival, and ultimately promote neurorecovery. This innovative strategy represents a paradigm shift, moving beyond mere protection to active cellular repair, offering a beacon of hope for stroke survivors.
Recharging Damaged Neurons: The Promise of Mitochondrial Transplantation
The concept of mitochondrial transplantation hinges on the idea that by providing fresh, functional mitochondria to cells starved of energy, we can rescue them from the brink of death and restore their metabolic capabilities. This approach is particularly compelling in the context of ischemic stroke, where mitochondrial dysfunction plays a central role in neuronal demise. Early research has demonstrated that transplanting autologous (self-donated) mitochondria into ischemic tissue can indeed mitigate injury. A recent Phase 1 trial has even begun to confirm the safety of autologous mitochondrial transplantation for acute cerebral ischemia in humans, marking a significant step forward in clinical translation Phase 1 trial confirms autologous transplant safety. This suggests that the body's own healthy mitochondria could be harvested and reintroduced to repair damaged brain cells, offering a personalized and potentially safer therapeutic route.
Navigating the Brain's Defenses: Delivery and Viability Challenges
Delivering therapeutic agents to the brain is notoriously challenging due to the blood-brain barrier. However, innovative strategies are being developed to overcome these hurdles for mitochondrial transplantation. Intraarterial delivery, for instance, has shown promise in effectively transporting mitochondria to the affected areas, leading to a significant reduction in cerebral infarction in preclinical models Intraarterial delivery significantly reduces cerebral infarction. Beyond delivery, ensuring the viability of transplanted mitochondria is paramount. Interestingly, studies indicate that standard periprocedural therapeutics, often used in stroke treatment, do not impair the extracellular viability of mitochondria during transplantation, which is crucial for maintaining their therapeutic potential Standard therapeutics maintain transplanted mitochondrial viability. This finding simplifies the clinical translation pathway, as existing treatment protocols may not need extensive modification to accommodate mitochondrial therapy.
Enhancing Repair Mechanisms: Scaffolds and Stem Cell Synergy
The therapeutic landscape for stroke recovery is constantly evolving, and mitochondrial transplantation is not operating in isolation. Researchers are exploring synergistic approaches to maximize its benefits. For example, combining mitochondrial transfer with stem cell therapies holds immense promise. Vascular endothelial growth factor-loaded hydrogel scaffolds, composed of chitosan and hyaluronic acid, have been shown to enhance the therapeutic effect of adipose-derived stem cells, specifically by boosting their capacity for mitochondrial transfer in the context of stroke Hydrogel scaffolds boost stem cell mitochondrial transfer. This suggests that creating an optimal microenvironment can amplify the regenerative power of these combined therapies. Furthermore, specific genetic modifications to stem cells, such as the combined deletion of Mitofusin 2, along with melatonin treatment, offer additional benefits in protecting the brain against acute ischemic stroke in rat models, primarily by enhancing mitochondrial neuroprotection Stem cell modifications enhance mitochondrial neuroprotection. These findings underscore the potential for multi-pronged strategies to achieve superior neurorestoration.
Tracking the Tiny Healers: Monitoring Mitochondrial Fate
For mitochondrial transplantation to become a mainstream therapy, understanding the fate and function of the transplanted organelles within the complex brain environment is critical. How long do they survive? Where do they go? How effectively do they integrate with host cells? Addressing these questions requires sophisticated tracking methods. A significant advancement in this area is the development of a first-in-field protocol for radiolabeling isolated mitochondria with Technetium-99m (Tc-99m) Novel radiolabeling enables tracking of transplanted mitochondria. This novel technique enables researchers to monitor the transplanted mitochondria non-invasively, providing invaluable insights into their biodistribution and persistence. Such tracking capabilities are essential for optimizing transplantation parameters, assessing therapeutic efficacy, and ensuring the safety of this emerging treatment. Moreover, ongoing research continues to unravel the intricate role of mitochondrial transmission in ischemic stroke, exploring whether it acts as a 'friend or foe' in regulating neurorecovery and inflammation Mitochondrial transmission regulates neurorecovery and inflammation, further highlighting the need for precise monitoring.
A Brighter Horizon for Stroke Recovery
Mitochondrial transplantation stands as a testament to the relentless pursuit of innovative solutions in neuroscience. By directly addressing the cellular energy crisis that underpins ischemic stroke, this therapy offers a profound shift from merely mitigating damage to actively promoting repair and regeneration. The progress from preclinical studies to early human trials, coupled with advancements in delivery, viability, synergistic approaches, and tracking technologies, paints a hopeful picture. As research continues to refine these techniques, we move closer to a future where stroke survivors can look forward to more complete and meaningful neurological recovery, ushering in a new era of restorative medicine for the brain.
Recent Posts
-
Mitochondrial Transplantation: A New Frontier in Restoring Neural Function After Ischemic Stroke
Imagine a future where the devastating impact of a stroke could be significantly mitigated, not jus …27th Mar 2026 -
Hijacking the Skull's Gateways: A New 'Backdoor' for Brain Drug Delivery
For decades, the brain has been considered an immune sanctuary, protected by the formidable blood-b …27th Mar 2026 -
CIDE: The New Bioinformatics Engine Unmasking Secreted Proteins for Precision Immunotherapy
For decades, the battle against cancer has focused on the cellular 'soldiers' and the genetic 'blue …27th Mar 2026