Description
HIV-1 p24 Residual Contamination PharmaGenie ELISA Kit
HIV-1 p24 is a core capsid protein of the human immunodeficiency virus that can be introduced as a process-related impurity when viral vectors or HIV-derived components are used in gene therapy, vaccine development, or other biopharmaceutical manufacturing processes. Even trace amounts of residual p24 antigen may indicate incomplete viral clearance, posing potential patient safety risks and regulatory non-compliance. Therefore, accurate measurement of p24 levels is a critical step in confirming product purity and ensuring adherence to stringent regulatory standards.
With its high sensitivity and specificity, the HIV-1 p24 Residual Contamination PharmaGenie ELISA Kit is a valuable tool for researchers and quality control teams working in gene therapy, vaccine production, and biopharmaceutical development.
| Product Name: | HIV-1 p24 Residual Contamination PharmaGenie ELISA Kit |
| Product Code: | AEGE00008 |
| Detection Method: | Sandwich ELISA |
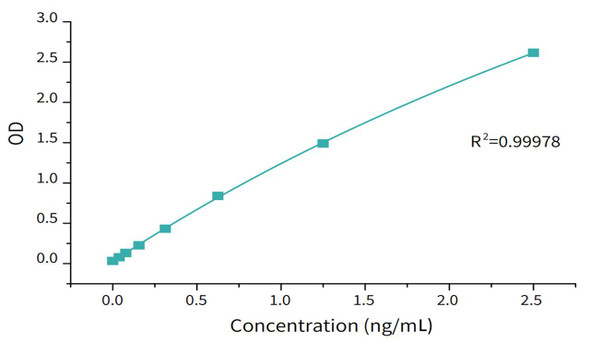
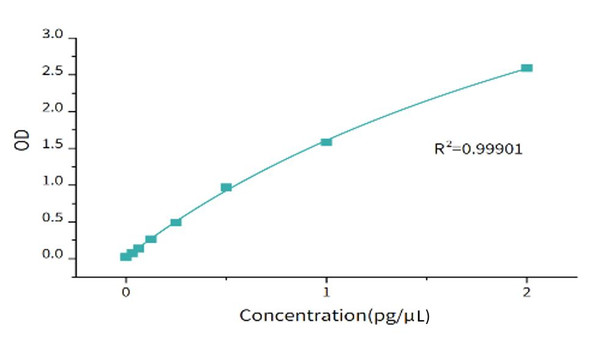
| Assay Range: | 6.25-200 pg/mL |
| Limit of Quantitation: | 6.25 pg/mL |
| Limit of Detection: | 3.125 pg/mL |
| Precision: | CV% ≤10%, RE% ≤ ±15% |
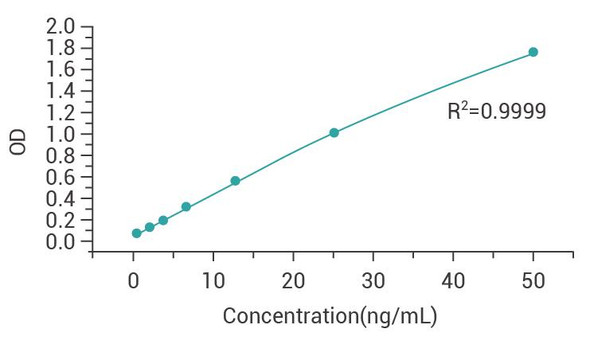
| Concentration of standard (pg/mL) | OD value 1 | OD value 2 | Mean value |
| 200 | 2.846 | 2.841 | 2.844 |
| 100 | 1.965 | 1.979 | 1.972 |
| 50 | 1.110 | 1.189 | 1.150 |
| 25 | 0.636 | 0.582 | 0.609 |
| 12.5 | 0.362 | 0.338 | 0.350 |
| 6.25 | 0.213 | 0.204 | 0.209 |
| 0 | 0.061 | 0.059 | 0.060 |