Aducanumab: Mechanism, Clinical Applications, and the Role of Biosimilars in Research
Quick Facts About Aducanumab
What is Aducanumab?
What is the mechanism of action for Aducanumab?
What are the clinical applications of Aducanumab?
Why is Aducanumab controversial?
1.) Understanding Aducanumab
Aducanumab is a monoclonal antibody developed by Biogen and Eisai that represents a major milestone in Alzheimer’s disease research. It was granted accelerated approval by the U.S. Food and Drug Administration (FDA) in 2021 due to its ability to reduce amyloid-beta plaques, a hallmark of Alzheimer’s pathology. However, its approval remains highly controversial, as clinical trial data did not provide definitive evidence of cognitive improvement. Instead, the decision was based on biomarker changes—specifically, the clearance of amyloid-beta from the brain—rather than conclusive proof of slowed disease progression. This has led to significant debate within the scientific and medical communities regarding its actual clinical benefit.
The introduction of Aducanumab has intensified interest in amyloid-targeting therapies, raising questions about their effectiveness in treating Alzheimer’s disease. While amyloid-beta accumulation is a recognized pathological feature, whether its removal translates into meaningful cognitive benefits remains uncertain. Clinical trials have yielded mixed results, with some studies indicating potential benefits and others failing to demonstrate consistent improvements. Moreover, concerns about safety, particularly the risk of amyloid-related imaging abnormalities (ARIA), including brain swelling and microhemorrhages, have necessitated cautious patient selection and monitoring strategies.
Despite these challenges, Aducanumab has paved the way for further research into Alzheimer’s treatments, influencing the development of next-generation anti-amyloid therapies such as Lecanemab and Donanemab. Ongoing studies aim to refine patient selection criteria, optimize dosing strategies, and explore combination approaches that integrate anti-amyloid therapies with other treatment modalities. As the field evolves, understanding the long-term effects of Aducanumab will be crucial in determining its role in Alzheimer’s management and shaping future therapeutic strategies.
2.) Mechanism of Action of Aducanumab
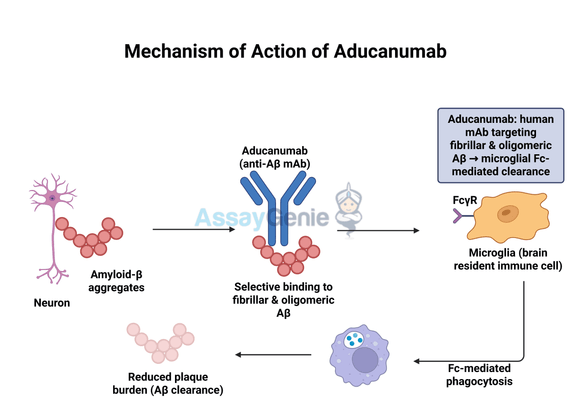
Aducanumab is a human monoclonal antibody designed to selectively target amyloid-beta aggregates, which are neurotoxic protein deposits implicated in Alzheimer’s disease progression. Unlike previous therapies that focused on symptom management, Aducanumab aims to modify disease pathology by directly reducing amyloid burden. Its mechanism of action involves several key processes:
- Selective Binding to Amyloid-Beta Aggregates: Aducanumab exhibits high affinity for fibrillar and oligomeric forms of amyloid-beta. These aggregates contribute to neurodegeneration by disrupting neuronal function and triggering inflammatory responses. By binding to these toxic forms, Aducanumab marks them for removal by the immune system.
- Microglial Activation and Plaque Clearance: Once Aducanumab binds to amyloid plaques, it engages microglia, the brain’s resident immune cells. Microglia play a central role in clearing cellular debris and misfolded proteins. Through Fc receptor-mediated phagocytosis, activated microglia engulf and degrade amyloid-beta deposits, leading to a measurable reduction in plaque burden as observed in clinical trials.
- Potential Neuroprotective Effects: The removal of amyloid-beta aggregates is hypothesized to mitigate downstream neurodegenerative processes, including synaptic dysfunction, neuroinflammation, and neuronal death. However, whether this translates into significant cognitive benefits remains a subject of ongoing investigation.
Despite its innovative approach, Aducanumab has been associated with adverse effects, particularly amyloid-related imaging abnormalities (ARIA). ARIA manifests as either edema (ARIA-E), characterized by temporary brain swelling, or microhemorrhages (ARIA-H), which involve small brain bleeds. These side effects have raised concerns about the safety of amyloid-targeting therapies, necessitating careful patient monitoring and individualized treatment approaches.
Further research is underway to refine Aducanumab’s clinical utility, optimize dosing regimens, and identify biomarkers that predict treatment response. Additionally, new anti-amyloid therapies, such as Lecanemab, aim to build upon Aducanumab’s approach while addressing its limitations, potentially improving efficacy and safety outcomes in Alzheimer’s patients.
3.) Clinical Applications of Aducanumab
Aducanumab is currently indicated for individuals with mild cognitive impairment (MCI) or early-stage Alzheimer’s disease. Its approval was largely based on clinical trials assessing its impact on amyloid-beta clearance and cognitive function. However, trial outcomes have been mixed, leading to ongoing discussions regarding its real-world effectiveness. Two pivotal studies, EMERGE and ENGAGE, played a crucial role in Aducanumab’s regulatory evaluation:
- EMERGE Trial: This study demonstrated that high-dose Aducanumab treatment led to a statistically significant reduction in cognitive decline compared to placebo. Patients receiving Aducanumab exhibited improvements in memory, orientation, and daily functioning, supporting its potential therapeutic benefit.
- ENGAGE Trial: In contrast, the ENGAGE trial failed to replicate these positive findings, showing no significant cognitive benefits in patients receiving Aducanumab. The discrepancies between the two studies fueled controversy, with critics arguing that the drug’s efficacy remains unproven.
Given the inconsistencies in clinical data, the approval of Aducanumab was met with skepticism from many experts. While some clinicians advocate for its use in select patients, others emphasize the need for more robust evidence demonstrating tangible cognitive improvements. As a result, Aducanumab’s role in Alzheimer’s treatment remains under reassessment, with ongoing studies aiming to clarify its long-term effects.
Beyond Aducanumab, newer anti-amyloid therapies such as Lecanemab and Donanemab are being developed to address the shortcomings of current treatments. These next-generation monoclonal antibodies aim to improve efficacy while minimizing side effects, offering hope for more effective Alzheimer’s interventions. Future research will focus on optimizing patient selection criteria, exploring combination therapies, and identifying novel treatment strategies that go beyond amyloid-beta clearance to address the multifaceted nature of Alzheimer’s disease.

4.) Exploring Biosimilars for Aducanumab
What is a Biosimilar?
| Aducanumab (Anti-APP) Biosimilar Antibody | |
|---|---|
| Antibody Type: | Monoclonal Antibody |
| Protein: | APP |
| Reactivity: | Human |
How Aducanumab Biosimilar Compares to Aducanumab
Benefits of Aducanumab Biosimilars in Research
- Cost-Effective: Enables broader research applications without the high cost of the original drug.
- Consistency in Studies: Provides standardized materials for preclinical and translational research.
- Potential for Future Innovations: Facilitates the development of next-generation Alzheimer’s treatments.
Research Use Only Disclaimer:
Discover Our Biosimilar Range
At Assay Genie, we specialize in providing high-quality biosimilars for research use! Check out our full biosimilar range to learn more.

By Marina Alberto, PhD
Marina Alberto, PhD, holds a robust academic background in Biotechnology, earning her Bachelor’s Degree and PhD in Science and Technology from Quilmes National University. Her research spans cancer immunotherapy, glycan profiling, and vaccine development, including innovative projects on pediatric leukemia diagnosis and cancer-associated carbohydrate-mimetic vaccines. She currently serves as a Technical Support and Sales Specialist at Assay Genie.
Recent Posts
-
Programmable Synthetic Hybrid Receptors (Hybrid-Rs): The Next Generation of Precision Immunotherapy
Imagine a world where cancer treatment is no longer a blunt instrument but a precision-guided surgi …31st Mar 2026 -
The Neural Shield: How Cancer-Induced Nerve Injury Drives Immunotherapy Resistance
For years, our understanding of cancer focused primarily on the tumor cells themselves and the immu …29th Mar 2026 -
Metabolic Bloodhounds: Engineering Immune Cells to Track Cancer's Chemical Trail
Imagine a specialized squad of immune cells, meticulously trained and equipped, not just to fight c …27th Mar 2026