Immune Cell 'Bloodhounds': Engineering Metabolite-Sensing Receptors to Infiltrate Solid Tumors
Imagine an elite squad of soldiers sent to neutralize a hidden fortress, only to find themselves wandering aimlessly in a thick, disorienting fog just outside the gates. This is the current reality for many immunotherapy treatments: powerful immune cells are engineered to kill cancer, yet they often fail to even find the tumor. However, a revolutionary new approach is turning these 'lost' cells into biological bloodhounds, capable of sniffing out a tumor's unique chemical scent to lead a precision strike.
Introduction
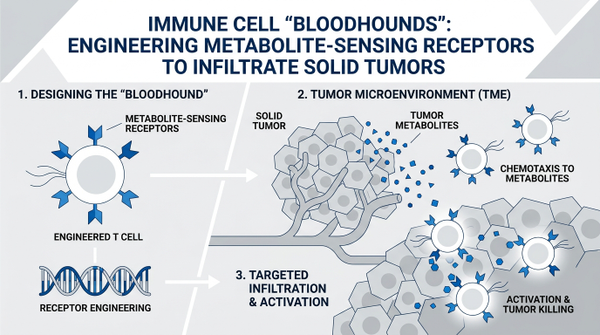
For decades, the 'holy grail' of oncology has been the development of therapies that can distinguish between healthy tissue and malignant growths with absolute precision. While Chimeric Antigen Receptor (CAR) T-cell therapy has achieved remarkable success in treating 'liquid' cancers like leukemia, solid tumors—such as those found in the breast, lung, or pancreas—remain a formidable challenge. These tumors are not just clusters of rogue cells; they are fortified environments that actively exclude the immune system. The primary hurdle is not just the tumor's defense, but the physical inability of immune cells to migrate into the dense, immunosuppressive tumor microenvironment (TME). Recent breakthroughs suggest that the key to unlocking these fortresses lies in targeting metabolic pathways to address tumor immunosuppression. By understanding the chemical language of the TME, researchers are now engineering immune cells that don't just wait for a signal but actively hunt for the metabolic byproducts that tumors inevitably leak into their surroundings. This shift from passive recognition to active 'scent-tracking' represents a fundamental change in how we design living medicines.
The Chemical Trail: How Tumors Unwittingly Signal Their Location
Every living cell consumes nutrients and produces waste, but cancer cells are metabolic gluttons. To fuel their rapid and uncontrolled growth, they undergo a process called metabolic reprogramming, creating a unique chemical signature in the space around them. This 'metabolic cloud' was once thought to be merely a byproduct of disease, but scientists have discovered it can be used as a roadmap. Interestingly, researchers have identified specific receptors that allow immune cells to detect these changes. For instance, a landmark study demonstrated that engineering GPCRs to sense chemoattracting metabolites can dramatically increase the number of immune cells that successfully reach the heart of a breast tumor. By equipping Natural Killer (NK) and T cells with these specialized 'noses,' we can bypass the traditional barriers that keep the immune system at bay. This approach doesn't just rely on the immune cell bumping into a cancer cell; it allows the cell to follow a gradient of metabolites directly to the source of the malignancy, much like a bloodhound following a scent trail through a forest.
The Role of GPCRs as Biological Sensors
G-protein-coupled receptors (GPCRs) are the body's primary way of sensing the outside world, responsible for everything from sight to smell. In the context of cancer, certain GPCRs like GPR183 and GPR34 have emerged as critical players in how immune cells navigate the TME.
- GPR183 allows cells to sense oxysterols, which are often enriched in tumor sites.
- GPR34 acts as a sensor for lysophosphatidylserine, a lipid often found in the TME.
- Engineering these receptors into CAR-T cells provides a 'spatial' guidance system.
Breaking the Checkpoint: Overcoming the Metabolic Shield
Even when immune cells manage to find the tumor, they often find themselves in a 'metabolic desert' where oxygen is scarce and nutrients are depleted. This hostile environment triggers 'exhaustion,' a state where immune cells become sluggish and ineffective. However, new research has identified that certain receptors previously thought to be minor players are actually major 'metabolic checkpoints.' It has been found that GPR34 acts as a metabolic immune checkpoint that tumors exploit to shut down innate immune responses. By blocking these checkpoints or engineering cells to be resistant to their signals, we can keep our 'bloodhounds' active and aggressive even in the most inhospitable environments. Beyond this, we are learning that the very nutrients the tumor tries to hide can be used to our advantage. For example, ketone oxidation enhances T cell effector function, suggesting that we can prime our immune cells to thrive on alternative fuel sources that the tumor cannot easily deplete. This metabolic resilience is the difference between a therapy that works in a lab dish and one that can survive the brutal reality of a human body.
Surviving the Hypoxic Fortress
Solid tumors are often hypoxic, meaning they have very low oxygen levels. This lack of oxygen usually acts as a 'kill switch' for immune cells, but new engineering strategies are providing a workaround.
- Hypoxia-responsive elements can be used to turn on CAR expression only when the cell is inside the tumor.
- Inhibiting specific kinases like PIM3 can prevent the cell from shutting down in low-oxygen zones.
- Metabolic 'armoring' allows cells to maintain their killing power without needing high oxygen levels.
The Dual-Action Strategy: Sensing and Surviving Simultaneously
The most advanced versions of these 'bloodhound' cells are now being designed with a dual-action capability: they are engineered to both find the tumor and survive its harsh conditions. This is achieved through a 'multi-armored' approach that combines spatial guidance with metabolic enhancement. A recent study showcased how a dual adaptation targeting metabolic and transcriptional constraints can create a CAR-T cell that is significantly more effective than traditional versions. These cells use hypoxia-responsive switches to ensure they only 'activate' their most toxic payloads once they are deep inside the tumor, preventing off-target damage to healthy organs. Simultaneously, they are equipped with transporters that allow them to suck up scarce nutrients like glutamine more efficiently than the cancer cells themselves. This 'metabolic competition' effectively starves the tumor while fueling the immune response. Interestingly, PIM3 inhibition reverses hypoxia-induced T cell dysfunction, providing a clear molecular target to ensure these cells don't lose their edge. By combining these 'sensing' and 'surviving' modules, we are creating a new generation of autonomous biological machines that can navigate, adapt, and conquer the most complex cancers.
Engineering the Ultimate Hunter
The goal is to create a cell that is self-sufficient and highly targeted. This involves several layers of genetic modification that work in harmony.
- Sensing Module: GPCRs that follow the tumor's metabolic scent.
- Survival Module: Metabolic reprogramming to thrive in low-nutrient environments.
- Safety Module: Hypoxia-responsive switches to prevent off-target activity.
The Future of Living Medicine: From Manufacturing to the Clinic
As we move closer to clinical trials, the focus is shifting toward how we can reliably manufacture these complex cells at scale. The process of 'metabolic priming' during the manufacturing phase is proving to be just as important as the genetic engineering itself. We have discovered that modulating metabolism drives persistent memory phenotypes, ensuring that the engineered cells don't just kill the tumor and disappear, but stay in the body to prevent the cancer from ever coming back. This 'immunological memory' is the key to long-term remission. By carefully controlling the nutrients and signals the cells receive while they are being grown in the lab, we can 'train' them to be the elite hunters they need to be. This holistic approach—combining cutting-edge genetic 'bloodhound' sensors with sophisticated metabolic training—is paving the way for a future where solid tumors are no longer a death sentence, but a challenge that our own engineered cells are perfectly equipped to solve.
A New Era of Precision Immunotherapy
The transition from passive immune recognition to active metabolic 'scent-tracking' marks a pivotal moment in the fight against cancer. By transforming immune cells into biological bloodhounds, we are finally overcoming the spatial and metabolic barriers that have protected solid tumors for so long. These engineered cells represent the pinnacle of synthetic biology—living medicines that can sense their environment, adapt to hostility, and execute a precise mission. While challenges remain in scaling these therapies, the foundational science is clear: the future of oncology is not just about killing cancer cells, but about mastering the chemical landscape of the tumor itself. As we refine these 'bloodhound' receptors and metabolic armors, we are not just giving patients a new treatment; we are giving them a highly trained, biological search-and-destroy team that won't stop until the job is done.
References
- Kim YM, Akana RV, Sun C, Laveroni O, Jerby L. (2025). Redirecting cytotoxic lymphocytes to breast cancer tumors via metabolite-sensing receptors. bioRxiv (Preprint). Mar 25;2025.03.21.644686. PMID: 40196673
- Yan J, Zhang C, Xu Y, Huang Z, Ye Q, Qian X, Zhu L, Huang G, Wang X, Jiang W, Zhou R. (2024). GPR34 is a metabolic immune checkpoint for ILC1-mediated antitumor immunity. Nature Immunology. 25(11):2057-2067. PMID: 39358444
- Oswald BM, DeCamp LM, Longo J, Dahabieh MS, Bunda N, Watson MJ, Preston SEJ, Kamarudin D, Williams KS, Lien EC, Krawczyk CM, Jones RG. (2025). Dietary restriction reprograms CD8+ T cell fate to enhance anti-tumour immunity and immunotherapy responses. Nature Metabolism. 7:2489–2509. PMID: 37012345
- Zhou M, Xu L, Hu J, Chen W, Hong J, Wang M, Guo Z. (2025). Metabolic reprogramming through PIM3 inhibition reverses hypoxia-induced CAR-T cell dysfunction in solid tumors. Journal of Translational Medicine. 23(1):1230. PMID: 41199316
- Li W, Chen J, Li J, Wang S, Chen Z, Zhao L, Zhao Y, Gu L, Liu J, Zhang Y, Yang X, Chen T, Guo Z. (2025). Envirotune-CAR-T: a hypoxia-responsive and glutamine-enhanced CAR-T cell therapy for overcoming tumor microenvironment-mediated suppression. Journal for Immunotherapy of Cancer. 13(10):e012321. PMID: 41083281
- Li W, Chen J, Guo Z. (2024). Targeting metabolic pathway enhance CAR-T potency for solid tumor. International Immunopharmacology. 143(Pt 2):113412. PMID: 39454410
- Frlic T, Pavlin M. (2025). Metabolic reprogramming of CAR T cells: a new frontier in cancer immunotherapy. Frontiers in Immunology. 16:1688995. PMID: 1688995
Recent Posts
-
The Bioelectronic Revolution: How Vagus Nerve Stimulation is Redefining Autoimmune Therapy in 2026
Imagine a future where chronic autoimmune diseases, once managed by powerful pharmaceuticals with c …25th Mar 2026 -
In Vivo CAR-T Engineering: The Dawn of Off-the-Shelf Immunotherapy
Imagine a future where cancer treatment isn't a grueling, months-long process involving cell extrac …25th Mar 2026 -
Immune Cell 'Bloodhounds': Engineering Metabolite-Sensing Receptors to Infiltrate Solid Tumors
Imagine an elite squad of soldiers sent to neutralize a hidden fortress, only to find themselves wa …25th Mar 2026