The Rise of Cancer Neuroscience: How Neural Circuits Drive Tumor Progression
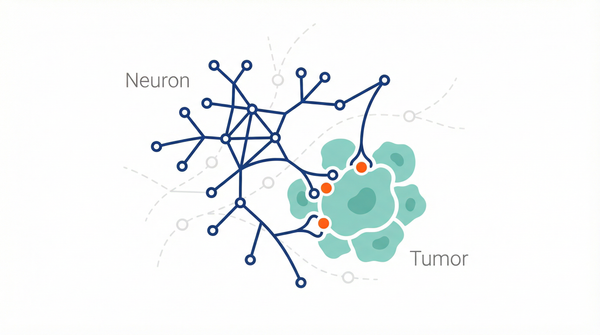
For decades, we viewed cancer as a rogue army of cells, a biological glitch driven solely by genetic mutations and metabolic greed. We imagined tumors as isolated fortresses, growing in a vacuum of their own making. But a radical shift is underway. Scientists have discovered that tumors are not just passive lumps of tissue; they are active participants in the body’s most complex communication network: the nervous system. This is the dawn of cancer neuroscience.
Introduction
The field of oncology is currently undergoing a profound transformation as we move beyond a purely cell-centric view of malignancy. Historically, the nervous system was thought to be a bystander in the oncogenic process, perhaps providing a structural scaffold but little else. However, groundbreaking research has revealed that the nervous system plays central role in cancer, acting as a master regulator of the tumor microenvironment. This realization has birthed the discipline of cancer neuroscience, which explores the intricate, bidirectional crosstalk between neural circuits and malignant cells. We now understand that tumors do not just grow near nerves; they actively recruit them, hijack their signaling pathways, and even form functional synapses with neurons to fuel their own progression. This neural dimension of cancer biology explains why some tumors are so resilient and why traditional therapies often fall short. By mapping these hidden connections, we are uncovering a new landscape of therapeutic vulnerabilities that could redefine how we treat the most aggressive forms of the disease.
The Malignant Synapse: How Gliomas Plug Into the Brain
Perhaps the most startling discovery in cancer neuroscience is that brain tumors don't just push neurons aside—they literally plug into them. High-grade gliomas have been found to form functional, electrochemical synapses with healthy neurons, allowing them to intercept and utilize the brain's own electrical signals. This isn't just a passive interaction; it is a sophisticated hijacking of the brain's fundamental machinery. Interestingly, these malignant synapses hijack adaptive plasticity mechanisms that are normally reserved for learning and memory. When a neuron fires, it releases neurotransmitters like glutamate, which the glioma cells receive through specialized receptors. This influx of electrical activity triggers a cascade of intracellular signals that drive the tumor to proliferate and invade deeper into the surrounding brain tissue. It is a vicious cycle where the brain’s own activity becomes the fuel for its destruction.
Hijacking Brain Plasticity
The tumor's ability to integrate into neural circuits is driven by factors like Brain-Derived Neurotrophic Factor (BDNF), which promotes the strengthening of these deadly connections. This process, known as malignant synaptic plasticity, mirrors the way our brains form new memories, but here it is repurposed to ensure the survival and expansion of the cancer.
- Formation of functional glutamatergic synapses between neurons and glioma cells.
- Activity-dependent depolarization of glioma membranes driving cell cycle progression.
- Recruitment of BDNF-TrkB signaling to enhance synaptic strength and tumor growth.
Long-Distance Influence: Remote Neurons Driving Infiltration
The influence of the nervous system on cancer isn't limited to the immediate vicinity of the tumor. Recent evidence suggests that neurons located far from the primary site can exert a powerful influence on tumor behavior. In the case of glioblastoma, researchers have observed that remote neuronal activity drives glioma infiltration across the brain's hemispheres. Specifically, activity in callosal projection neurons—which connect the two halves of the brain—can stimulate the migration of tumor cells into previously healthy regions. This finding is revolutionary because it suggests that the entire brain's network state, not just the local environment, dictates the pace of cancer spread. It explains the frustratingly diffuse nature of certain brain cancers, where cells seem to appear far from the original mass almost overnight.
The SEMA4F Signaling Axis
The mechanism behind this long-distance drive involves the axon guidance molecule SEMA4F. When remote neurons are active, they trigger signaling pathways that encourage glioma cells to adopt a highly mobile, invasive phenotype. This highlights the importance of considering the brain as a whole when designing surgical and radiological interventions.
The Sympathetic Rewiring of the Tumor Microenvironment
Beyond the brain, the peripheral nervous system is equally involved in the cancer narrative. The sympathetic nervous system, responsible for our 'fight or flight' response, has been shown to infiltrate solid tumors and fundamentally alter their surroundings. It appears that sympathetic nerves rewire the tumor microenvironment to create a sanctuary for cancer cells. By releasing norepinephrine and other signaling molecules, these nerves can suppress immune cell activity, promote the formation of new blood vessels (angiogenesis), and even alter the stiffness of the extracellular matrix. This neural remodeling makes the tumor more resistant to chemotherapy and more likely to metastasize to distant organs. It is as if the cancer is using the body's own stress response system to shield itself from harm and pave the way for its expansion.
Neural Sabotage: How Nerve Injury Blunts Immunotherapy
One of the most significant hurdles in modern oncology is the development of resistance to immunotherapy. We are now learning that the nervous system may be a key culprit in this process. As tumors grow, they often invade and damage local nerves, a process known as perineural invasion. This nerve injury promotes immunotherapy resistance mechanisms by triggering a chronic inflammatory response. The injured nerves release a cocktail of cytokines, such as IL-6, which effectively 'exhaust' the T cells that are supposed to attack the tumor. Instead of a robust immune response, the microenvironment becomes a site of immune suppression. This discovery is particularly vital because it suggests that by protecting nerves or blocking the signals they release when injured, we might be able to restore the effectiveness of life-saving immunotherapies like anti-PD-1 treatments.
The Unique Challenges of the Metastatic Brain Landscape
When cancer spreads from other parts of the body to the brain, it encounters a uniquely hostile yet strangely accommodating environment. The brain's blood-brain barrier and its specialized immune cells, the microglia, create a distinct set of rules for survival. Research into brain metastasis exhibits unique organotropic mechanisms that allow circulating tumor cells to cross into the neural sanctuary and thrive. These metastatic cells must learn to 'speak the language' of the brain, often adopting neural-like characteristics to avoid detection and tap into local nutrient sources. Understanding these specific adaptations is crucial for developing targeted therapies that can prevent or treat brain metastases, which remain one of the most lethal complications of advanced cancer.
A New Frontier in Precision Oncology
The rise of cancer neuroscience marks a turning point in our battle against malignancy. By recognizing that tumors are integrated into the body's neural architecture, we are opening a vast new frontier for therapeutic intervention. We are no longer just targeting the cancer cell; we are targeting the conversation between the nerve and the tumor. Whether it is through pharmacological blockade of malignant synapses, protecting nerves from injury-induced inflammation, or disrupting the sympathetic rewiring of the microenvironment, the possibilities are profound. As we continue to decode the neural code of cancer, we move closer to a future where precision oncology encompasses the full complexity of human biology, offering new hope to patients with the most challenging diagnoses.
References
- Huang-Hobbs E, Cheng YT, Ko Y, et al. (2023). Remote neuronal activity drives glioma progression through SEMA4F. Nature. 619(7971):844-850. PMID: 37380778
- Taylor KR, Barron T, Hui A, et al. (2023). Glioma synapses recruit mechanisms of adaptive plasticity. Nature. 623(7986):366-374. PMID: 37914930
- Winkler F, Venkatesh HS, Amit M, et al. (2023). Cancer neuroscience: State of the field, emerging directions. Cell. 186(8):1689-1707. PMID: 37059069
- Sattler A, Korzun T, Gupta K, et al. (2025). Sympathetic nerve signaling rewires the tumor microenvironment: a shift in "microenvironmental-ity". Cancer Metastasis Rev. 44(1):25. PMID: 39831934
- Baruch EN, Gleber-Netto FO, Nagarajan P, et al. (2025). Cancer-induced nerve injury promotes resistance to anti-PD-1 therapy. Nature. 646(8084):462-473. PMID: 40836096
- Valiente M, Anders C, Boire A, et al. (2026). The evolving landscape of brain metastasis: volume II. Trends Cancer. 12(2):116-146. PMID: 41339184
23rd Feb 2026
Recent Posts
-
Turning Tumors Against Themselves: In Vivo Reprogramming of Cancer Cells into Dendritic Sentinels
Imagine a future where cancer cells, once masters of disguise, are forced to reveal themselves and …3rd Apr 2026 -
Programmable Synthetic Hybrid Receptors (Hybrid-Rs): The Next Generation of Precision Immunotherapy
Imagine a world where cancer treatment is no longer a blunt instrument but a precision-guided surgi …31st Mar 2026 -
The Neural Shield: How Cancer-Induced Nerve Injury Drives Immunotherapy Resistance
For years, our understanding of cancer focused primarily on the tumor cells themselves and the immu …29th Mar 2026