Emapalumab: Unlocking the Potential of Targeting IFN-γ in HLH Treatment
Quick Facts About Emapalumab
What is Emapalumab?
What is the mechanism of action of Emapalumab?
What are the clinical applications of Emapalumab?
1.) Understanding Emapalumab
Emapalumab represents a groundbreaking advancement in the treatment of hemophagocytic lymphohistiocytosis (HLH), a life-threatening hyperinflammatory disorder characterized by excessive immune activation and uncontrolled cytokine release. HLH is a rare but severe condition that leads to systemic inflammation, multi-organ failure, and, if untreated, high mortality rates. Traditional treatment options have primarily relied on broad immunosuppressive therapies, including corticosteroids, etoposide, and hematopoietic stem cell transplantation (HSCT). However, these approaches often present significant limitations, including high toxicity, incomplete disease control, and severe immunosuppression, which can leave patients vulnerable to life-threatening infections.
Developed by Novimmune and later acquired by Swedish Orphan Biovitrum (Sobi), Emapalumab is the first and only FDA-approved monoclonal antibody specifically designed to target and neutralize interferon-gamma (IFN-γ), a key cytokine implicated in HLH pathogenesis. IFN-γ plays a central role in driving excessive macrophage activation, leading to widespread inflammation and tissue damage. By selectively inhibiting IFN-γ, Emapalumab provides a targeted approach to restoring immune balance without the widespread immunosuppression associated with conventional therapies.
The approval of Emapalumab marked a paradigm shift in HLH management, significantly improving survival outcomes in patients who fail to respond to standard treatments. Beyond primary HLH, ongoing research is evaluating its potential in secondary HLH, macrophage activation syndrome (MAS), and other inflammatory disorders driven by IFN-γ dysregulation. Given its precision-targeted mechanism, Emapalumab has emerged as a transformative therapy, offering a more effective and safer alternative to traditional immunosuppressive regimens.
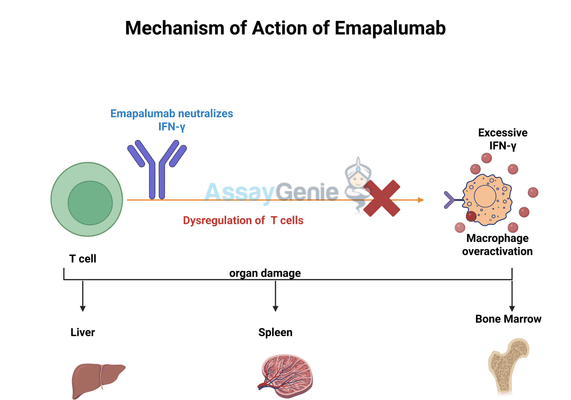
2.) Mechanism of Action of Emapalumab
The pathophysiology of HLH is driven by an unchecked immune response, where dysregulated T cells and natural killer (NK) cells fail to regulate macrophage activity, leading to a massive cytokine storm. IFN-γ is a pivotal cytokine in this process, as it amplifies inflammatory signaling, sustains macrophage hyperactivation, and perpetuates the uncontrolled immune response. The resulting excessive inflammation causes severe tissue damage in critical organs, including the liver, spleen, and bone marrow, further complicating HLH treatment.
Emapalumab exerts its therapeutic effects by binding to soluble IFN-γ, neutralizing its activity, and breaking the inflammatory cycle that drives HLH progression. The key effects of Emapalumab include:
- Suppressing excessive immune activation: By blocking IFN-γ, Emapalumab prevents further macrophage overactivation, which is a hallmark of HLH. This targeted inhibition helps restore immune regulation without completely shutting down protective immune responses.
- Reducing tissue inflammation: The drug mitigates inflammation-related organ damage by dampening IFN-γ-driven inflammatory signaling in key tissues. This effect is particularly beneficial in patients experiencing HLH-associated liver dysfunction, splenomegaly, or bone marrow suppression.
This precision-targeted mechanism makes Emapalumab a particularly valuable therapeutic option for HLH patients who do not respond adequately to conventional therapies. Furthermore, emerging research suggests that IFN-γ blockade may offer benefits in other hyperinflammatory conditions, such as macrophage activation syndrome (MAS), cytokine release syndrome (CRS), and severe viral infections. By addressing the underlying cytokine dysregulation in these conditions, Emapalumab holds promise as a broader immunomodulatory therapy for diseases driven by excessive IFN-γ signaling.
3.) Clinical Applications of Emapalumab
Primary HLH: The First FDA-Approved Treatment
Emapalumab became the first FDA-approved treatment for primary HLH in 2018, revolutionizing disease management for this rare and often fatal immune disorder. By targeting IFN-γ, it directly addresses the hyperinflammatory state characteristic of HLH, significantly improving survival rates in patients refractory to conventional therapies. Clinical trials demonstrated that Emapalumab reduced cytokine-driven inflammation and increased remission rates before HSCT, minimizing transplant-related complications and offering a targeted alternative for high-risk patients.
Secondary HLH and Macrophage Activation Syndrome (MAS)
While approved for primary HLH, Emapalumab also holds promise for secondary HLH, triggered by infections, malignancies, or autoimmune diseases. Both conditions share excessive IFN-γ production, and preliminary studies suggest that Emapalumab could effectively control inflammation in secondary HLH. Similarly, MAS, a severe inflammatory complication in autoimmune diseases like sJIA and SLE, may also benefit from IFN-γ blockade, particularly in patients unresponsive to corticosteroids.
Beyond HLH and MAS, Emapalumab is being explored for cytokine-mediated disorders, including severe viral infections (e.g., EBV-associated HLH, COVID-19), cytokine release syndrome (CRS) from CAR-T therapy, and graft-versus-host disease (GVHD). Ongoing trials aim to expand its clinical applications, potentially establishing Emapalumab as a key therapeutic for IFN-γ-driven inflammatory diseases. With its targeted mechanism, Emapalumab continues to shape the future of immunotherapy, offering new hope for patients with severe immune dysregulation.

4.) Exploring Biosimilars for Emapalumab
What is a Biosimilar?
| Emapalumab (Anti-IFNG) Biosimilar Antibody | |
|---|---|
| Antibody Type: | Monoclonal Antibody |
| Protein: | IFNG |
| Reactivity: | Human |
How Does an Emapalumab Biosimilar Compare to the Original Drug?
Advancing Research on Emapalumab
Biosimilars play a crucial role in expanding immunotherapy research. By providing access to high-quality, research-use-only alternatives, biosimilars enable:
- Preclinical and translational studies: Researchers can investigate new applications of IFN-γ blockade.
- Mechanistic studies: Biosimilars allow for in-depth exploration of immune modulation in inflammatory diseases.
- Drug combination testing: Studying biosimilars in combination with other therapies can uncover synergistic treatment strategies.
Research Use Only Disclaimer:
Discover Our Biosimilar Range
At Assay Genie, we specialize in providing high-quality biosimilars for research use! Check out our full biosimilar range to learn more.

By Chris McNally, PhD
Chris McNally, PhD, has a strong foundation in Biomedical Science, completing a PhD scholarship in collaboration with Randox Laboratories and Ulster University. Chris has published extensively in prostate cancer research, focusing on biomarker discovery, cancer risk stratification, and molecular mechanisms such as hypoxia-induced regulation. He currently serves as a Business Development Manager at Assay Genie.
Recent Posts
-
IgG1 Plasma Cells: The Emerging Biomarker for Predicting Cancer Immunotherapy Success
In the relentless fight against cancer, immunotherapy has emerged as a beacon of hope, harnessing t …24th Feb 2026 -
The Rise of Cancer Neuroscience: How Neural Circuits Drive Tumor Progression
For decades, we viewed cancer as a rogue army of cells, a biological glitch driven solely by geneti …23rd Feb 2026 -
CRISPR-Powered Light Sensors: A New Frontier in Ultra-Sensitive Cancer Detection
Cancer detection often relies on advanced imaging or invasive procedures, frequently catching the d …20th Feb 2026