Description
Human Ferritin light chain (FTL) ELISA Kit
The Human Ferritin Light Chain (FTL) ELISA Kit is specifically designed for the precise measurement of FTL levels in human serum, plasma, and cell culture supernatants. This kit offers exceptional sensitivity and accuracy, guaranteeing consistent and dependable results for various research purposes.FTL is a key protein involved in iron metabolism, serving as a vital component in the storage and transportation of iron in the body. Abnormal FTL levels have been linked to various health conditions, including anemia, hemochromatosis, and inflammatory disorders, making it a valuable biomarker for studying these diseases and advancing potential treatments.
With its advanced technology and superior performance, the Human Ferritin Light Chain (FTL) ELISA Kit is an essential tool for researchers and clinicians seeking to explore the role of FTL in iron-related disorders and develop targeted interventions for improved patient outcomes.
| Product Name: | Human Ferritin light chain (FTL) ELISA Kit |
| SKU: | HUEB0045 |
| Size: | 96T |
| Target: | Human Ferritin light chain (FTL) |
| Synonyms: | Ferritin L subunit |
| Assay Type: | Sandwich |
| Detection Method: | ELISA |
| Reactivity: | Human |
| Detection Range: | 31.2-2000pg/mL |
| Sensitivity: | 10pg/mL |
| Intra CV: | 3.6% | ||||||||||||||||||||
| Inter CV: | 7.9% | ||||||||||||||||||||
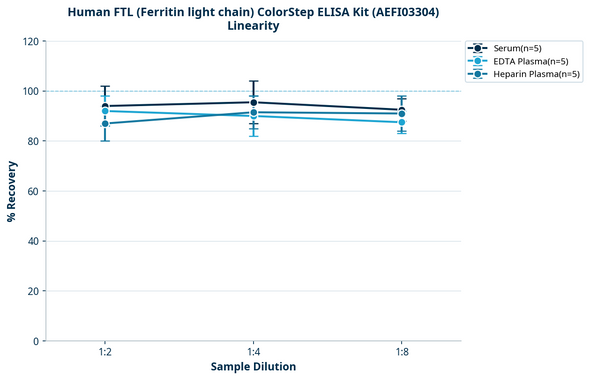
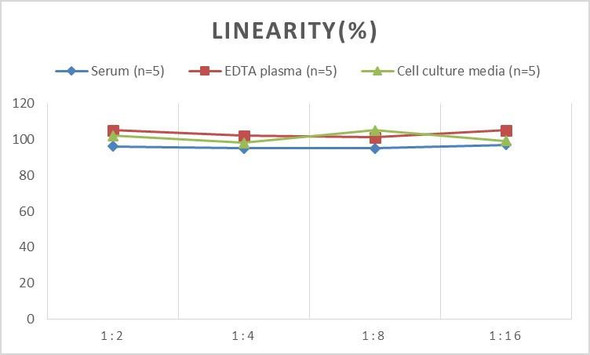
| Linearity: |
| ||||||||||||||||||||
| Recovery: |
| ||||||||||||||||||||
| Function: | Stores iron in a soluble, non-toxic, readily available form. Important for iron homeostasis. Iron is taken up in the ferrous form and deposited as ferric hydroxides after oxidation. Also plays a role in delivery of iron to cells. Mediates iron uptake in capsule cells of the developing kidney. |
| Uniprot: | P02792 |
| Sample Type: | Serum, plasma, tissue homogenates, cell culture supernates and other biological fluids |
| Specificity: | Natural and recombinant human Ferritin light chain |
| Sub Unit: | Oligomer of 24 subunits. There are two types of subunits: L (light) chain and H (heavy) chain. The major chain can be light or heavy, depending on the species and tissue type. The functional molecule forms a roughly spherical shell with a diameter of 12 nm and contains a central cavity into which the insoluble mineral iron core is deposited. Iron enters the spherical protein shell through pores that are formed between subunits. Mutations leading to truncation or the addition of extra residues at the C-terminus interfere with normal pore formation and with iron accumulation. |
| Research Area: | Neurosciences |
| Storage: | Please see kit components below for exact storage details |
| Note: | For research use only |
| UniProt Protein Function: | FTL: Stores iron in a soluble, non-toxic, readily available form. Important for iron homeostasis. Iron is taken up in the ferrous form and deposited as ferric hydroxides after oxidation. Also plays a role in delivery of iron to cells. Mediates iron uptake in capsule cells of the developing kidney. Defects in FTL are the cause of hereditary hyperferritinemia-cataract syndrome (HHCS). It is an autosomal dominant disease characterized by early-onset bilateral cataract. Affected patients have elevated level of circulating ferritin. HHCS is caused by mutations in the iron responsive element (IRE) of the FTL gene. Defects in FTL are the cause of neurodegeneration with brain iron accumulation type 3 (NBIA3); also known as adult-onset basal ganglia disease. It is a movement disorder with heterogeneous presentations starting in the fourth to sixth decade. It is characterized by a variety of neurological signs including parkinsonism, ataxia, corticospinal signs, mild nonprogressive cognitive deficit and episodic psychosis. It is linked with decreased serum ferritin levels. Belongs to the ferritin family. |
| UniProt Protein Details: | Protein type:Oxidoreductase Chromosomal Location of Human Ortholog: 19q13.33 Cellular Component: ferritin complex; membrane; cytosol Molecular Function:identical protein binding; protein binding; ferric iron binding; iron ion binding Biological Process: receptor-mediated endocytosis; iron ion homeostasis; cellular iron ion homeostasis; post-Golgi vesicle-mediated transport; iron ion transport; transmembrane transport Disease: Hyperferritinemia With Or Without Cataract; L-ferritin Deficiency; Neurodegeneration With Brain Iron Accumulation 3 |
| NCBI Summary: | This gene encodes the light subunit of the ferritin protein. Ferritin is the major intracellular iron storage protein in prokaryotes and eukaryotes. It is composed of 24 subunits of the heavy and light ferritin chains. Variation in ferritin subunit composition may affect the rates of iron uptake and release in different tissues. A major function of ferritin is the storage of iron in a soluble and nontoxic state. Defects in this light chain ferritin gene are associated with several neurodegenerative diseases and hyperferritinemia-cataract syndrome. This gene has multiple pseudogenes. [provided by RefSeq, Jul 2008] |
| UniProt Code: | P02792 |
| NCBI GenInfo Identifier: | 120523 |
| NCBI Gene ID: | 2512 |
| NCBI Accession: | P02792.2 |
| UniProt Secondary Accession: | P02792,Q6IBT7, Q7Z2W1, Q86WI9, Q8WU07, Q96AU9, Q96CU0 Q9BTZ8, B2R4B9, |
| UniProt Related Accession: | P02792 |
| Molecular Weight: | 440 kDa |
| NCBI Full Name: | Ferritin light chain |
| NCBI Synonym Full Names: | ferritin, light polypeptide |
| NCBI Official Symbol: | FTL |
| NCBI Official Synonym Symbols: | LFTD; NBIA3 |
| NCBI Protein Information: | ferritin light chain; ferritin L-chain; ferritin L subunit; ferritin light polypeptide-like 3 |
| UniProt Protein Name: | Ferritin light chain |
| Protein Family: | Ferritin |
| UniProt Gene Name: | FTL |
| UniProt Entry Name: | FRIL_HUMAN |
| Component | Quantity (96 Assays) | Storage |
| ELISA Microplate (Dismountable) | 8×12 strips | -20°C |
| Lyophilized Standard | 2 | -20°C |
| Sample Diluent | 20ml | -20°C |
| Assay Diluent A | 10mL | -20°C |
| Assay Diluent B | 10mL | -20°C |
| Detection Reagent A | 120µL | -20°C |
| Detection Reagent B | 120µL | -20°C |
| Wash Buffer | 30mL | 4°C |
| Substrate | 10mL | 4°C |
| Stop Solution | 10mL | 4°C |
| Plate Sealer | 5 | - |
Other materials and equipment required:
- Microplate reader with 450 nm wavelength filter
- Multichannel Pipette, Pipette, microcentrifuge tubes and disposable pipette tips
- Incubator
- Deionized or distilled water
- Absorbent paper
- Buffer resevoir
*Note: The below protocol is a sample protocol. Protocols are specific to each batch/lot. For the correct instructions please follow the protocol included in your kit.
Allow all reagents to reach room temperature (Please do not dissolve the reagents at 37°C directly). All the reagents should be mixed thoroughly by gently swirling before pipetting. Avoid foaming. Keep appropriate numbers of strips for 1 experiment and remove extra strips from microtiter plate. Removed strips should be resealed and stored at -20°C until the kits expiry date. Prepare all reagents, working standards and samples as directed in the previous sections. Please predict the concentration before assaying. If values for these are not within the range of the standard curve, users must determine the optimal sample dilutions for their experiments. We recommend running all samples in duplicate.
| Step | |
| 1. | Add Sample: Add 100µL of Standard, Blank, or Sample per well. The blank well is added with Sample diluent. Solutions are added to the bottom of micro ELISA plate well, avoid inside wall touching and foaming as possible. Mix it gently. Cover the plate with sealer we provided. Incubate for 120 minutes at 37°C. |
| 2. | Remove the liquid from each well, don't wash. Add 100µL of Detection Reagent A working solution to each well. Cover with the Plate sealer. Gently tap the plate to ensure thorough mixing. Incubate for 1 hour at 37°C. Note: if Detection Reagent A appears cloudy warm to room temperature until solution is uniform. |
| 3. | Aspirate each well and wash, repeating the process three times. Wash by filling each well with Wash Buffer (approximately 400µL) (a squirt bottle, multi-channel pipette,manifold dispenser or automated washer are needed). Complete removal of liquid at each step is essential. After the last wash, completely remove remaining Wash Buffer by aspirating or decanting. Invert the plate and pat it against thick clean absorbent paper. |
| 4. | Add 100µL of Detection Reagent B working solution to each well. Cover with the Plate sealer. Incubate for 60 minutes at 37°C. |
| 5. | Repeat the wash process for five times as conducted in step 3. |
| 6. | Add 90µL of Substrate Solution to each well. Cover with a new Plate sealer and incubate for 10-20 minutes at 37°C. Protect the plate from light. The reaction time can be shortened or extended according to the actual color change, but this should not exceed more than 30 minutes. When apparent gradient appears in standard wells, user should terminatethe reaction. |
| 7. | Add 50µL of Stop Solution to each well. If color change does not appear uniform, gently tap the plate to ensure thorough mixing. |
| 8. | Determine the optical density (OD value) of each well at once, using a micro-plate reader set to 450 nm. User should open the micro-plate reader in advance, preheat the instrument, and set the testing parameters. |
| 9. | After experiment, store all reagents according to the specified storage temperature respectively until their expiry. |
When carrying out an ELISA assay it is important to prepare your samples in order to achieve the best possible results. Below we have a list of procedures for the preparation of samples for different sample types.
| Sample Type | Protocol |
| Serum | If using serum separator tubes, allow samples to clot for 30 minutes at room temperature. Centrifuge for 10 minutes at 1,000x g. Collect the serum fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. If serum separator tubes are not being used, allow samples to clot overnight at 2-8°C. Centrifuge for 10 minutes at 1,000x g. Remove serum and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. |
| Plasma | Collect plasma using EDTA or heparin as an anticoagulant. Centrifuge samples at 4°C for 15 mins at 1000 × g within 30 mins of collection. Collect the plasma fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. Note: Over haemolysed samples are not suitable for use with this kit. |
| Urine & Cerebrospinal Fluid | Collect the urine (mid-stream) in a sterile container, centrifuge for 20 mins at 2000-3000 rpm. Remove supernatant and assay immediately. If any precipitation is detected, repeat the centrifugation step. A similar protocol can be used for cerebrospinal fluid. |
| Cell culture supernatant | Collect the cell culture media by pipette, followed by centrifugation at 4°C for 20 mins at 1500 rpm. Collect the clear supernatant and assay immediately. |
| Cell lysates | Solubilize cells in lysis buffer and allow to sit on ice for 30 minutes. Centrifuge tubes at 14,000 x g for 5 minutes to remove insoluble material. Aliquot the supernatant into a new tube and discard the remaining whole cell extract. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Tissue homogenates | The preparation of tissue homogenates will vary depending upon tissue type. Rinse tissue with 1X PBS to remove excess blood & homogenize in 20ml of 1X PBS (including protease inhibitors) and store overnight at ≤ -20°C. Two freeze-thaw cycles are required to break the cell membranes. To further disrupt the cell membranes you can sonicate the samples. Centrifuge homogenates for 5 mins at 5000xg. Remove the supernatant and assay immediately or aliquot and store at -20°C or -80°C. |
| Tissue lysates | Rinse tissue with PBS, cut into 1-2 mm pieces, and homogenize with a tissue homogenizer in PBS. Add an equal volume of RIPA buffer containing protease inhibitors and lyse tissues at room temperature for 30 minutes with gentle agitation. Centrifuge to remove debris. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Breast Milk | Collect milk samples and centrifuge at 10,000 x g for 60 min at 4°C. Aliquot the supernatant and assay. For long term use, store samples at -80°C. Minimize freeze/thaw cycles. |