Macrophage, M1 & M2, Polarization and Function
What is a Macrophage?
Macrophages are indeed a type of phagocyte, responsible for engulfing and destroying foreign particles and cellular debris. They are larger cells with more cytoplasm and are found in various tissues throughout the body, where they serve as part of the innate immune response.. They play a crucial role in maintaining the body's defense mechanism by engulfing and digesting cellular debris, foreign substances, pathogens, and even cancer cells. This process, known as phagocytosis, involves the internalization of external agents into the macrophage cell.
Apart from their phagocytic abilities, macrophages also act as potent signaling hubs in the immune system. They release inflammatory molecules and mediators called cytokines, which serve as messengers for other immune cells, orchestrating a coordinated response to infections. Notable examples of these cytokines include TNF (tumor necrosis factor), IL-1, IL-6, IL-8, and IL-12.
Additionally, macrophages actively contribute to combating infections by producing antimicrobial mediators like complement proteins and reactive oxygen species. These antimicrobial agents effectively eliminate the pathogens that have been engulfed during phagocytosis. It's important to note that macrophages are derived from blood monocytes, which are non-specialized cells of the innate immune system found in the bone marrow. The differentiation of monocytes into macrophages occurs in response to specific stimuli encountered at the site of infection.
Key Takeaways
- Macrophages are key immune cells that engulf pathogens and secrete cytokines, playing a vital role in defense and tissue repair.
- They exist in various forms, including M1 and M2 subtypes, each with distinct functions in inflammation and healing.
What do marcophages secrete?
Macrophages, a vital type of white blood cell, serve as key players in the immune response, playing a pivotal role in combating bacterial and viral infections. One of their primary functions is phagocytosis, wherein they engulf and neutralize harmful microorganisms. As a result of this process, macrophages release essential signaling molecules called cytokines and chemokines.
Cytokines, a class of proteins secreted by macrophages, help regulate and coordinate the immune response. Simultaneously, chemokines act as chemoattractants, guiding other immune cells to the site of infection, bolstering the body's defense. These macrophage-derived cytokines and chemokines can be categorized into three main groups: pro-inflammatory, anti-inflammatory, and regulatory. Pro-inflammatory cytokines and chemokines initiate the inflammatory response, a crucial defense mechanism to combat infections. Conversely, anti-inflammatory counterparts play a role in resolving the inflammatory process and promoting healing.
Furthermore, regulatory cytokines and chemokines modulate the overall immune response, ensuring a balanced and controlled reaction to foreign invaders. The significance of macrophage-derived cytokines and chemokines lies in their dual role: orchestrating the initiation and resolution of the immune response. By functioning as essential mediators in the immune system, macrophage-derived cytokines and chemokines play a key role in safeguarding the body from infections. Their contributions are paramount in maintaining a robust immune defense and promoting overall health.
Macrophage Polarization by T-helper Cells
Morphology of Macrophages
Studying macrophage morphology is achieved through various staining techniques, which involve using specific dyes to highlight different aspects of these cells, facilitating their observation under a microscope.
One commonly employed staining technique is Wright-Giemsa staining, where a combination of dyes imparts a purple or blue color to macrophages. This method is particularly useful for examining the structure of macrophage cell membranes. On the other hand, May-Grunwald Giemsa staining employs a different mixture of dyes, resulting in macrophages appearing pink or red. Researchers often use this method to investigate the internal structure of macrophages, including the location of their nucleus and other organelles.
Regardless of the staining technique employed, the study of macrophage morphology provides valuable insights into the mechanisms by which these cells defend the body against diseases.
M1 & M2 Macrophages
M1 and M2 macrophages are two different functional states or phenotypes of macrophages. These states represent the diverse roles that macrophages can play in the immune response and tissue homeostasis. The designations "M1" and "M2" are often used to simplify the classification of macrophages based on their functions, but in reality, macrophages can exhibit a spectrum of activities and characteristics that may not fit neatly into these categories.
M1 macrophages are often referred to as "classically activated" macrophages. They are typically induced by pro-inflammatory signals, such as interferon-gamma (IFN-gamma) and microbial products like lipopolysaccharide (LPS). M1 macrophages have a potent ability to produce pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-alpha), interleukin-1 beta (IL-1beta), and interleukin-6 (IL-6). They are efficient at killing pathogens, promoting inflammation, and initiating immune responses against infections or foreign invaders. M1 macrophages also play a role in promoting tissue damage in certain inflammatory conditions.
M2 macrophages are often referred to as "alternatively activated" macrophages. They can be induced by anti-inflammatory signals, such as interleukin-4 (IL-4), interleukin-13 (IL-13), or immune complexes. M2 macrophages have anti-inflammatory properties and are involved in tissue repair, remodeling, and resolution of inflammation. They produce anti-inflammatory cytokines, such as interleukin-10 (IL-10) and transforming growth factor-beta (TGF-beta), which help dampen inflammation and promote tissue healing. M2 macrophages also play a role in regulating immune responses and promoting tissue regeneration.
M2b macrophages, sometimes known as regulatory macrophages (Mregs), are characterized by producing significant amounts of the anti-inflammatory cytokine interleukin-10 (IL-10) and relatively low levels of the pro-inflammatory cytokine interleukin-12 (IL-12). M2b macrophages are involved in regulating immune responses and promoting tolerance.
M2c macrophages, often referred to as deactivated or resolution-phase macrophages, release substantial quantities of IL-10 and another anti-inflammatory cytokine, transforming growth factor-beta (TGF-β). These macrophages are essential for dampening inflammation, promoting tissue repair, and contributing to the resolution of immune responses after an infection or injury. M2c macrophages are induced by glucocorticoids (a class of steroid hormones with anti-inflammatory properties) and TGF-β, which can be released in response to anti-inflammatory signals.
M2d macrophages are a subtype with pro-angiogenic properties, meaning they promote the growth of new blood vessels. These macrophages secrete IL-10, TGF-β, and vascular endothelial growth factor (VEGF), a key factor in stimulating angiogenesis. M2d macrophages are activated by the A2 adenosine receptor agonist IL-6 (A2R).
Macrophage ELISA Kits
Related ELISA Kits
Cluster of Differentiation 163 (CD163) is a protein found on the surface of natural killer cells, which play an important role in fighting cancer cells and infected cells. CD163 also functions in the activation and proliferation of T cells. CD163 is used as a marker for the differentiation of Th1 cells

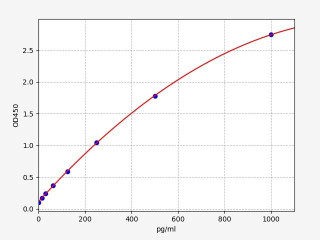
| Human CD80/B7-1 PharmaGenie ELISA - Kit Data | |
|---|---|
| ELISA Type | Sandwich |
| Sensitivity | 12 pg/ml |
| Range | 12 pg/ml - 3000 pg/ml |
The antibody pair provided in this kit recognizes human B7-1 / CD80.
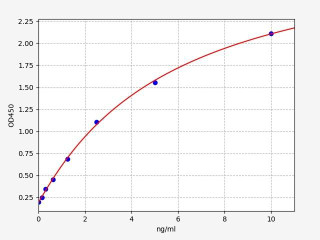
| Human CD86 / B7-2 ELISA KitA | |
|---|---|
| ELISA Type | Sandwich |
| Sensitivity | 00.094ng/ml |
| Range | 0.156-10ng/ml |
CD86 (CD86 Molecule) is a type I transmembrane glycoprotein and belongs to the B7 family. It is also known as CD86 antigen or BB1. CD86 is expressed on the surface of activated B cells, T cells, dendritic cells, macrophages and some epithelial cells. It binds to its receptor (CD28) on T cells, which leads to the activation of these cells. The interaction between CD86 and CD28 is important for the initiation and regulation of immune responses.
Macrophage Function
Macrophages play a critical role in identifying pathogens by utilizing pattern recognition receptors (PRRs), specialized molecules that target specific components on the surface of these intruders. Among these receptors, toll-like receptors (TLRs) are notably present on the surface of macrophages.
Upon recognizing distinct molecules like LPS, peptidoglycan, or flagellin on the surface of pathogens, the macrophages initiate phagocytosis of these invaders. Subsequently, they release an array of pro-inflammatory molecules that play a pivotal role in recruiting diverse immune cells to the infection site, facilitating the removal of the foreign organisms.
Macrophages exhibit a remarkable ability to migrate and circulate through almost every tissue, perpetually scanning for pathogens and eliminating dead cells. Their differentiation is influenced by the tissue they inhabit, enabling them to adapt to specific microenvironments. The following information outlines various types of specialized macrophages found in different tissues.
Alveolar macrophages
Alveolar macrophages, a subtype of lung macrophages, reside within the alveoli of the lungs. These specialized macrophages play a crucial role in eliminating disease-causing agents, toxic substances, and allergens that manage to bypass the respiratory tract's mechanical defenses. In addition to their defensive functions, alveolar macrophages actively contribute to preserving lung health by clearing away deceased and dying cells, as well as other cellular debris.
Tumour associated macrophages (TAMs)
Tumor-associated macrophages (TAMs) assume a pivotal role in facilitating tumor metastasis through the establishment of an immunosuppressive tumor microenvironment (TME). Within this context, TAMs orchestrate the production of cytokines, chemokines, and growth factors. Additionally, they trigger the release of inhibitory immune checkpoint proteins within T cells. This intricate interplay culminates in the attenuation of T cell function, consequently fostering an environment conducive to tumor progression and metastasis. TAMs are a distinct subset of macrophages that tumors recruit into the TME to engineer an immunosuppressive milieu, allowing the tumor to evade immune surveillance and thrive.
Macrophages in the Neuroblastoma Tumour Microenvironment
Aggressive neuroblastoma (NB) frequently manifests a notably vascular and immunosuppressive tumor microenvironment (TME) primarily inhabited by macrophages. This setting facilitates the transformation of these macrophages into "tumor-associated macrophages" (TAMs), characterized by a distinct M2 phenotypic and functional profile.
M2 macrophages play a pivotal role in orchestrating several crucial anti-inflammatory functions, including tissue repair, angiogenesis, and fibrosis, especially during instances of tissue injury. However, their involvement can become pathogenic in the context of tumor development and progression. These macrophages often contribute to adverse processes such as tumor invasion, degradation of the extracellular matrix, and suppression of the adaptive immune system.
CD86 macrophage
One of the molecules involved in costimulating T cells is called CD86, also known as B7-2. CD86 is a type I membrane protein that is a member of the immunoglobulin superfamily. It is expressed on antigen-presenting cells (including macrophages, B cells, and DCs) to provide the costimulatory signals necessary for T cell activation.
When a macrophage encounters a pathogen, it will phagocytose the pathogen and then present pieces of it (antigens) on its surface. CD86 is expressed on the surface of macrophages and helps to deliver the costimulatory signals that activate T cells. T cells then help to coordinate the immune response and eliminate the pathogen.
Without CD86, macrophages would be unable to properly costimulate T cells and the immune response would be compromised. Therefore, CD86 is an essential molecule for macrophage function and overall immune system.
Macrophages in atherosclerosis
Macrophages play an important role in atherosclerotic lesions. They actively participate in lipoprotein ingestion and accumulation, giving rise to foam cells filled with lipid droplets. The accumulation of foam cells contributes to lipid storage and atherosclerotic plaque growth. Macrophages are important in the development and progression of atherosclerosis and can be a target for therapeutic interventions.
Macrophage Polarization
There are two main types of macrophage: M1 and M2. M1 macrophages are pro-inflammatory and help to fight infection, while M2 macrophages are anti-inflammatory and help to promote healing.
Macrophages can become polarized into either an M1 or M2 state in response to different stimuli. For example, exposure to bacterial products or cytokines (signaling molecules produced by immune cells) will trigger the M1 polarization, while exposure to viruses or glucocorticoids (steroid hormones that are released in response to stress) will trigger the M2 polarization.
Once a macrophage has been polarized into an M1 or M2 state, it will remain in that state for the rest of its life. This is because the M1 and M2 states are characterized by different gene expression patterns. M1 macrophages, for example, express high levels of pro-inflammatory cytokines such as IL-6 and TNF-alpha, while M2 macrophages express high levels of anti-inflammatory cytokines such as IL-10 and IL-1RA.
The M1 and M2 states are not mutually exclusive. Macrophages can exist in a mixed state, where they express both M1 and M2 genes. However, they will usually be skewed towards one state or the other depending on the stimuli they are exposed to.
In conclusion, macrophages represent integral components of the immune milieu, exerting multifaceted influences across diverse biological contexts. Their vigilant surveillant activity within tissues, coupled with their intricate immune-regulatory and phagocytic functions, underscores their pivotal role in maintaining immune homeostasis. Furthermore, macrophages' capacity to discern and internalize various substrates, alongside their nuanced involvement in shaping tumor microenvironments, highlights their dynamic and versatile attributes. A comprehensive grasp of macrophage functionality not only augments our comprehension of immunological intricacies but also unveils prospects for innovative therapeutic avenues across a spectrum of pathological scenarios.
Written by Pragna Krishnapur
Pragna Krishnapur completed her bachelor degree in Biotechnology Engineering in Visvesvaraya Technological University before completing her masters in Biotechnology at University College Dublin.
Recent Posts
-
ELISA vs Western Blot: Which Technique Should You Choose?
Written by Seán Mac Fhearraigh, PhD • Updated: 19 May 2026 • ~9 min read Quick Answer ELISA …20th May 2026 -
Types of ELISA: Direct, Indirect, Sandwich & Competitive Compared
Written by Seán Mac Fhearraigh, PhD • Updated: 19 May 2026 • ~9 min read Quick Answer The fi …20th May 2026 -
Sandwich ELISA: Step-by-Step Protocol & Troubleshooting
Written by Seán Mac Fhearraigh, PhD • Updated: 19 May 2026 • ~ …20th May 2026