Description
Sheep Metabolic Stress ELISA Sampler Pack
The Sheep Metabolic Stress ELISA Sampler Pack from Assay Genie is a comprehensive set of validated ELISA kits designed to support sheep metabolic stress research. Antioxidant enzymes and lipid peroxidation markers for ovine oxidative stress and nutritional research. Each kit in the pack delivers reliable and reproducible quantification of its target analyte across serum, plasma, and other relevant sheep biological samples.
By combining these analytes in a single sampler pack, researchers can efficiently profile multiple biomarkers relevant to metabolic stress, accelerating biomarker discovery, mechanistic studies, and translational research in sheep models. Order the Sheep Oxidative Stress ELISA Sampler Pack today to streamline your multi-analyte sheep workflow.
Sheep Metabolic Stress ELISA Sampler Pack
The Sheep Metabolic Stress ELISA Sampler Pack from Assay Genie allows researchers to begin their investigation into sheep metabolic stress via analysing GPx, Catalase, MDA and SOD in a 96 assay format per kit, thus allowing researchers to identify and validate analytes within the pathway.
| Pack Contents | Size | Reactivity | Sensitivity | Range | ELISA Type | Datasheet | MSDS |
| Sheep GPX1 (Glutathione Peroxidase) ELISA Kit | 96 Assays | Sheep | 0.094 ng/mL | 0.156-10 ng/mL | Sandwich ELISA | Datasheet | MSDS |
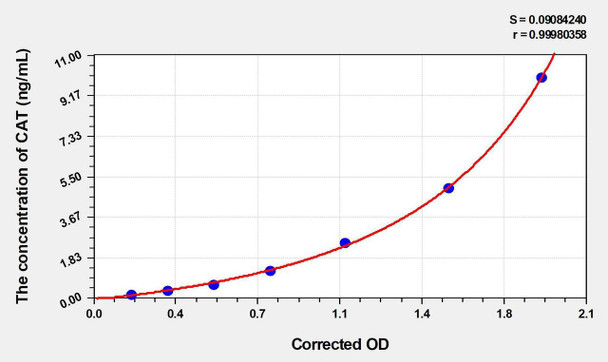
| Sheep CAT (Catalase) ELISA Kit | 96 Assays | Sheep | 0.052 ng/mL | 0.16-10 ng/mL | Sandwich ELISA | Datasheet | MSDS |
| Sheep MDA (Malondialdehyde) ELISA Kit | 96 Assays | Sheep | 9.02 ng/mL | 31.25-2000 ng/mL | Competitive ELISA | Datasheet | MSDS |
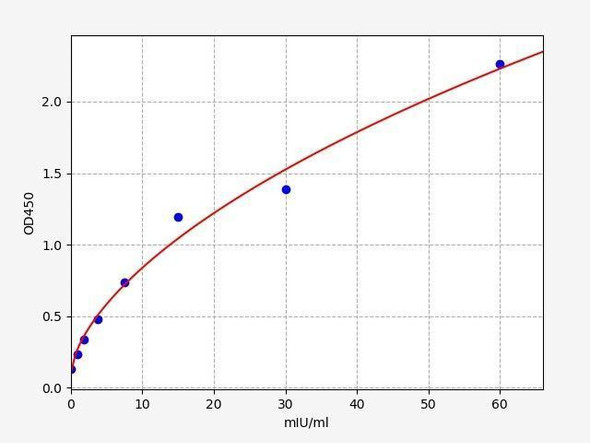
| Sheep SOD (Superoxide Dismutases) ELISA Kit | 96 Assays | Sheep | 0.066 U/mL | 0.16-10 U/mL | Sandwich ELISA | Datasheet | MSDS |
This Sampler Pack contains both Sandwich and Competitive ELISA kits. Use the tabs below to view the components included with each kit type.
| Component | Quantity | Storage |
| ELISA Microplate (Dismountable) | 4-12 strips | 4°C for 6 months |
| Lyophilized Standard | 1 | 4°C/-20°C |
| Sample/Standard Dilution Buffer | 10ml | 4°C |
| Biotin-labeled Antibody(Concentrated) | 60ul | 4°C (Protect from light) |
| Antibody Dilution Buffer | 5ml | 4°C |
| HRP-Streptavidin Conjugate(SABC) | 60ul | 4°C (Protect from light) |
| SABC Dilution Buffer | 5ml | 4°C |
| TMB Substrate | 10ml | 4°C (Protect from light) |
| Stop Solution | 5ml | 4°C |
| Wash Buffer(25X) | 15ml | 4°C |
| Plate Sealer | 3 | - |
Other materials and equipment required:
- Microplate reader with 450 nm wavelength filter
- Multichannel Pipette, Pipette, microcentrifuge tubes and disposable pipette tips
- Incubator
- Deionized or distilled water
- Absorbent paper
- Buffer resevoir
| Component | Quantity (24 Assays) | Quantity (96 Assays) | Storage |
| Micro ELISA Plate (Dismountable) | 8 wells × 3 strips | 8 wells × 12 strips | -20°C, 6 months |
| Reference Standard | 1 vial | 2 vials | |
| Concentrated Biotinylated Detection Ab (100×) | 1 vial, 60 µL | 1 vial, 120 µL | |
| Concentrated HRP Conjugate (100×) | 1 vial, 60 µL | 1 vial, 120 µL | -20°C (shading light), 6 months |
| Reference Standard & Sample Diluent | 1 vial, 20 mL | 1 vial, 20 mL | 4°C, 6 months |
| Biotinylated Detection Ab Diluent | 1 vial, 14 mL | 1 vial, 14 mL | |
| HRP Conjugate Diluent | 1 vial, 14 mL | 1 vial, 14 mL | |
| Concentrated Wash Buffer (25×) | 1 vial, 30 mL | 1 vial, 30 mL | |
| Substrate Reagent | 1 vial, 10 mL | 1 vial, 10 mL | 4°C (shading light) |
| Stop Solution | 1 vial, 10 mL | 1 vial, 10 mL | 4°C |
| Plate Sealer | 5 pieces | 5 pieces | |
| Product Description | 1 copy | 1 copy |
Other materials and equipment required:
- Microplate reader with 450 nm wavelength filter
- Multichannel Pipette, Pipette, microcentrifuge tubes and disposable pipette tips
- Incubator at 37°C
- Deionized or distilled water
- Absorbent paper
- Buffer reservoir
When carrying out an ELISA assay it is important to prepare your samples in order to achieve the best possible results. Below we have a list of procedures for the preparation of samples for different sample types.
| Sample Type | Protocol |
| Serum | If using serum separator tubes, allow samples to clot for 30 minutes at room temperature. Centrifuge for 10 minutes at 1,000x g. Collect the serum fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. If serum separator tubes are not being used, allow samples to clot overnight at 2-8°C. Centrifuge for 10 minutes at 1,000x g. Remove serum and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. |
| Plasma | Collect plasma using EDTA or heparin as an anticoagulant. Centrifuge samples at 4°C for 15 mins at 1000 - g within 30 mins of collection. Collect the plasma fraction and assay promptly or aliquot and store the samples at -80°C. Avoid multiple freeze-thaw cycles. Note: Over haemolysed samples are not suitable for use with this kit. |
| Urine & Cerebrospinal Fluid | Collect the urine (mid-stream) in a sterile container, centrifuge for 20 mins at 2000-3000 rpm. Remove supernatant and assay immediately. If any precipitation is detected, repeat the centrifugation step. A similar protocol can be used for cerebrospinal fluid. |
| Cell culture supernatant | Collect the cell culture media by pipette, followed by centrifugation at 4°C for 20 mins at 1500 rpm. Collect the clear supernatant and assay immediately. |
| Cell lysates | Solubilize cells in lysis buffer and allow to sit on ice for 30 minutes. Centrifuge tubes at 14,000 x g for 5 minutes to remove insoluble material. Aliquot the supernatant into a new tube and discard the remaining whole cell extract. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Tissue homogenates | The preparation of tissue homogenates will vary depending upon tissue type. Rinse tissue with 1X PBS to remove excess blood & homogenize in 20ml of 1X PBS (including protease inhibitors) and store overnight at ≤ -20°C. Two freeze-thaw cycles are required to break the cell membranes. To further disrupt the cell membranes you can sonicate the samples. Centrifuge homogenates for 5 mins at 5000xg. Remove the supernatant and assay immediately or aliquot and store at -20°C or -80°C. |
| Tissue lysates | Rinse tissue with PBS, cut into 1-2 mm pieces, and homogenize with a tissue homogenizer in PBS. Add an equal volume of RIPA buffer containing protease inhibitors and lyse tissues at room temperature for 30 minutes with gentle agitation. Centrifuge to remove debris. Quantify total protein concentration using a total protein assay. Assay immediately or aliquot and store at ≤ -20 °C. |
| Milk | Collect milk samples and centrifuge at 10,000 x g for 60 min at 4°C. Aliquot the supernatant and assay. For long term use, store samples at -80°C. Minimize freeze/thaw cycles. |
This Sampler Pack contains both Sandwich and Competitive ELISA kits, which follow different reaction protocols. Use the tabs below to view the relevant protocol for each kit type.
*Note: Protocols are specific to each batch/lot. For the correct instructions please follow the protocol included in your kit.
Before adding to wells, equilibrate the SABC working solution and TMB substrate for at least 30 min at 37°C. When diluting samples and reagents, they must be mixed completely and evenly. It is recommended to plot a standard curve for each test.
| Step | Protocol |
| 1. | Set standard, test sample and control (zero) wells on the pre-coated plate respectively, and then, record their positions. It is recommended to measure each standard and sample in duplicate. Wash plate 2 times before adding standard, sample and control (zero) wells! |
| 2. | Aliquot 0.1ml standard solutions into the standard wells. |
| 3. | Add 0.1 ml of Sample / Standard dilution buffer into the control (zero) well. |
| 4. | Add 0.1 ml of properly diluted sample into test sample wells. |
| 5. | Seal the plate with a cover and incubate at 37 °C for 90 min. |
| 6. | Remove the cover and discard the plate content, clap the plate on the absorbent filter papers or other absorbent material. Do NOT let the wells completely dry at any time. Wash plate X2. |
| 7. | Add 0.1 ml of Biotin- detection antibody working solution into the above wells (standard, test sample & zero wells). Add the solution at the bottom of each well without touching the side wall. |
| 8. | Seal the plate with a cover and incubate at 37°C for 60 min. |
| 9. | Remove the cover, and wash plate 3 times with Wash buffer. Let wash buffer rest in wells for 1 min between each wash. |
| 10. | Add 0.1 ml of SABC working solution into each well, cover the plate and incubate at 37°C for 30 min. |
| 11. | Remove the cover and wash plate 5 times with Wash buffer, and each time let the wash buffer stay in the wells for 1-2 min. |
| 12. | Add 90 µl of TMB substrate into each well, cover the plate and incubate at 37°C in dark within 10-20 min. |
| 13. | Add 50 µl of Stop solution into each well and mix thoroughly. The color changes into yellow immediately. |
| 14. | Read the O.D. absorbance at 450 nm in a microplate reader immediately after adding the stop solution. |
*Note: Protocols are specific to each batch/lot. For the correct instructions please follow the protocol included in your kit.
In a Competitive ELISA, the target analyte in the sample competes with a fixed amount of pre-coated antigen for binding to the biotinylated detection antibody. Higher analyte concentrations therefore produce a LOWER signal (inverse relationship). Before starting, equilibrate all reagents to room temperature for at least 30 min and prepare working dilutions per the supplied datasheet.
| Step | Protocol |
| 1. | Set standard, blank, and sample wells on the pre-coated plate. Measure standards and samples in duplicate. Record well positions. |
| 2. | Add 50 µl of standard or sample to the appropriate wells. Add 50 µl of standard / sample diluent to the blank wells. |
| 3. | Immediately add 50 µl of the Biotinylated Detection Antibody working solution into each well. Gently shake to mix. |
| 4. | Cover the plate with a sealer and incubate at 37°C for 45 min. |
| 5. | Remove the sealer and aspirate / decant the well content. Wash the plate 3 times with 1× Wash Buffer (350 µl per well), letting the buffer soak for 1 min between washes. Pat dry on absorbent paper. |
| 6. | Add 100 µl of HRP Conjugate working solution into each well. Cover and incubate at 37°C for 30 min. |
| 7. | Aspirate / decant and wash the plate 5 times as in step 5. |
| 8. | Add 90 µl of Substrate Reagent to each well. Cover and incubate at 37°C, protected from light, for 15 min (or until the most concentrated standard wells develop a deep blue colour). |
| 9. | Add 50 µl of Stop Solution to each well. The colour will change from blue to yellow. |
| 10. | Immediately read the O.D. absorbance at 450 nm using a microplate reader. Plot a 4-parameter logistic standard curve (note the inverse relationship for competitive ELISA) and calculate sample concentrations. |