Eldelumab: Unlocking the Potential of Anti-TNFSF14 in Inflammatory Diseases
Quick Facts About Eldelumab
What is Eldelumab?
How does Eldelumab work?
What are the clinical applications of Eldelumab?
1.) Understanding Eldelumab
Eldelumab is a human monoclonal antibody developed to inhibit Tumor Necrosis Factor Superfamily Member 14 (TNFSF14), also known as LIGHT. This cytokine plays a crucial role in immune signaling, influencing T-cell activation, cytokine production, and interactions between immune cells. TNFSF14’s dysregulation has been linked to chronic inflammatory and autoimmune conditions, making it an attractive target for novel immunotherapies.
Originally developed for treating autoimmune diseases, Eldelumab has been investigated primarily in ulcerative colitis (UC) and Crohn’s disease (CD), both of which involve persistent gastrointestinal inflammation driven by an overactive immune response. Preclinical and early clinical studies suggest that TNFSF14 blockade may help regulate excessive immune activation, reduce tissue damage, and improve symptoms in these conditions. However, while promising, Eldelumab has not yet received regulatory approval for widespread clinical use.
Beyond inflammatory bowel disease (IBD), researchers are exploring its potential applications in other autoimmune disorders such as rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE), where LIGHT signaling contributes to disease progression. Unlike traditional immunosuppressive therapies, which broadly dampen immune function, Eldelumab’s targeted mechanism offers a more selective approach, potentially reducing side effects while maintaining immune defense mechanisms.
Despite these advantages, challenges remain in optimizing its therapeutic application. Clinical trials have yielded mixed results, necessitating further research to refine patient selection criteria, dosage regimens, and combination strategies with other immunomodulatory agents. As investigations continue, Eldelumab remains a key candidate in the evolving landscape of immune-targeted therapies, offering new hope for patients with treatment-resistant autoimmune and inflammatory diseases.
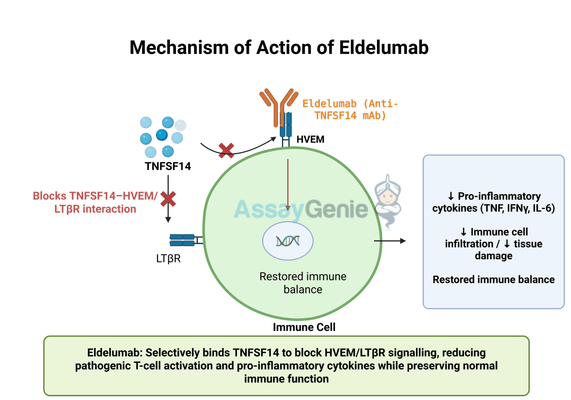
2.) Mechanism of Action of Eldelumab
Eldelumab functions by selectively binding to TNFSF14, preventing its interaction with its primary receptors, Herpesvirus Entry Mediator (HVEM) and Lymphotoxin Beta Receptor (LTβR). These receptors are widely expressed on immune cells and play a crucial role in regulating immune activation, inflammation, and tissue remodeling.
Under normal conditions, TNFSF14 signaling contributes to immune homeostasis by promoting T-cell activation and cytokine production. However, in autoimmune diseases, excessive TNFSF14 activity leads to chronic inflammation, increased immune cell infiltration, and tissue destruction. By inhibiting TNFSF14, Eldelumab effectively disrupts this pathological immune response, helping to restore immune balance and reduce inflammation.
One of the key advantages of targeting TNFSF14 is its specificity in modulating immune responses without completely suppressing immune function. Unlike broad-spectrum immunosuppressants that can lead to increased infection risk, Eldelumab’s selective inhibition may provide a safer alternative for managing autoimmune diseases. Additionally, its mechanism may complement existing therapies, enhancing treatment efficacy when used in combination with other immunomodulatory agents.
Preclinical studies and early clinical trials have demonstrated Eldelumab’s potential in reducing pro-inflammatory cytokine levels and limiting immune cell recruitment in affected tissues. This has been particularly relevant in conditions such as IBD, where sustained inflammation leads to chronic gastrointestinal damage. However, ongoing research aims to further elucidate the long-term effects of TNFSF14 inhibition, optimize dosing strategies, and identify biomarkers that predict patient response.
Despite promising early results, challenges remain in translating these findings into clinical success. Researchers continue to refine Eldelumab’s therapeutic applications, ensuring that its benefits outweigh potential risks and that it can be effectively integrated into existing treatment regimens.
3.) Clinical Applications of Eldelumab
Eldelumab has been primarily investigated for its potential in treating chronic inflammatory and autoimmune diseases, with a focus on ulcerative colitis and Crohn’s disease. Both conditions involve excessive immune activation in the gastrointestinal tract, leading to persistent inflammation, mucosal damage, and debilitating symptoms. By targeting TNFSF14, Eldelumab offers a novel approach to modulating immune responses in IBD, potentially reducing disease severity and improving patient outcomes.
Clinical trials evaluating Eldelumab in ulcerative colitis and Crohn’s disease have provided valuable insights into its therapeutic potential. Early studies suggest that inhibiting TNFSF14 may help reduce inflammatory cytokine production, limit immune cell infiltration in intestinal tissues, and promote mucosal healing. However, results have been mixed, with some trials showing only modest benefits, highlighting the need for further research to optimize patient selection and combination strategies.
Beyond IBD, Eldelumab’s mechanism suggests broader applications in autoimmune diseases where TNFSF14 plays a role. Rheumatoid arthritis, systemic lupus erythematosus, and psoriasis are among the conditions being explored as potential indications. Given the role of TNFSF14 in T-cell regulation, Eldelumab may also have implications in transplant medicine, where controlling immune responses is crucial for preventing graft rejection.
Despite its promise, challenges remain in clinical development. Determining the most responsive patient populations, refining dosing strategies, and assessing long-term safety are key areas of ongoing research. Additionally, researchers are exploring how Eldelumab could be integrated with other therapies, including biologics and small-molecule inhibitors, to enhance treatment efficacy.
As investigations continue, Eldelumab represents a potential breakthrough in immune-targeted therapy, offering new hope for patients with treatment-resistant autoimmune diseases and inflammatory conditions.

4.) Exploring Biosimilars for Eldelumab
What is a Biosimilar?
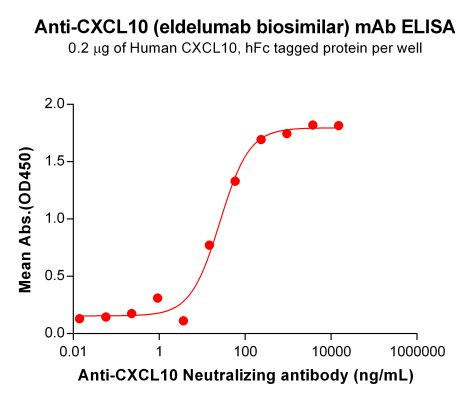
| Eldelumab (Anti-CXCL10) Biosimilar Antibody | |
|---|---|
| Antibody Type: | Monoclonal Antibody |
| Protein: | CXCL10 |
| Reactivity: | Human |
Comparison Between Eldelumab and Eldelumab Biosimilar
Benefits of Eldelumab Biosimilar in Research
- Accessibility: Biosimilars increase the availability of investigational drugs for preclinical and translational research.
- Cost-Effectiveness: More affordable options for studying TNFSF14 inhibition in immune-mediated diseases.
- Regulatory Flexibility: Suitable for research purposes, allowing further exploration of TNFSF14’s role in immune modulation.
Research Use Only Disclaimer:
Discover Our Biosimilar Range
At Assay Genie, we specialize in providing high-quality biosimilars for research use! Check out our full biosimilar range to learn more.

By Miren Ruiz de Eguilaz, PhD
Miren Ruiz de Eguilaz, PhD, has an extensive academic background, earning a BSc in Biology from UPV/EHU, an MSc in Biotechnology from the University of Oviedo, and a PhD in Chemistry from Dublin City University (DCU). Miren’s expertise lies in biosensor technology and bacterial diagnostics. She currently serves as a Product Manager at Assay Genie.
Recent Posts
-
IgG1 Plasma Cells: The Emerging Biomarker for Predicting Cancer Immunotherapy Success
In the relentless fight against cancer, immunotherapy has emerged as a beacon of hope, harnessing t …24th Feb 2026 -
The Rise of Cancer Neuroscience: How Neural Circuits Drive Tumor Progression
For decades, we viewed cancer as a rogue army of cells, a biological glitch driven solely by geneti …23rd Feb 2026 -
CRISPR-Powered Light Sensors: A New Frontier in Ultra-Sensitive Cancer Detection
Cancer detection often relies on advanced imaging or invasive procedures, frequently catching the d …20th Feb 2026