Understanding Immunoglobulin Antibodies: Structure, Function, and Types
Immunoglobulin antibodies stand at the forefront of the immune defense system, offering a sophisticated mechanism for recognizing and neutralizing a vast array of pathogens, including bacteria, viruses, and other potentially harmful foreign bodies. These antibodies, also known as immunoglobulins (Ig), are produced by specialized cells in the immune system and serve as a critical component of both innate and adaptive immunity. This article provides a deeper understanding of the structure, function, and classification of immunoglobulin antibodies, highlighting their essential role in maintaining health and combating diseases.
Detailed Structure of Immunoglobulin Antibodies
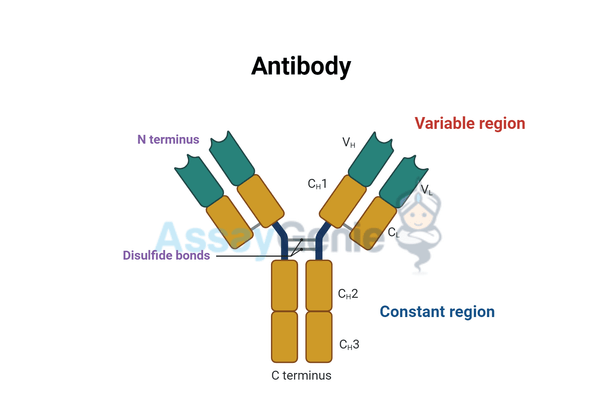
At the molecular level, immunoglobulin antibodies are Y-shaped molecules composed of four polypeptide chains: two identical heavy chains and two identical light chains, linked together by disulfide bonds. The structure of an antibody can be further divided into two functional regions:
1. Fab Region (Antigen-binding Fragment)
Location of the Fab Region
The Arms of the Y-shaped Molecule
- Positioning: The Fab region constitutes the upper parts of the antibody's Y-shaped structure, extending outwards to allow interaction with antigens.
- Composition: Each arm of the antibody contains one Fab region, which is further divided into a variable (V) region at the tip and a constant (C) region closer to the antibody's trunk.
Function of the Fab Region
The functions of the Fab (Fragment, antigen-binding) region of an immunoglobulin antibody include:
Antigen Recognition: Specifically identifies and binds to antigens.
High Specificity: Due to its variable (V) domain, it can recognize a wide variety of antigens with high specificity.
Neutralization: Prevents pathogens from infecting cells by binding to them.
2. Fc Region (Crystallizable Fragment)
The Fc (Fragment, crystallizable) region, integral to the functionality and specificity of immunoglobulin antibodies, serves as the backbone for the diverse roles antibodies play within the immune system. Here's an expanded overview of its location and function:
Location of the Fc Region
- Stem of the Y-Shape: The Fc region forms the base or stem of the antibody's Y-shaped structure. Unlike the variable Fab regions that bind antigens, the Fc region remains constant within each class of antibodies, providing a stable scaffold that mediates interactions with other immune components.
Function of Fc Region
Immune Cell Activation: It binds to Fc receptors on immune cells, triggering phagocytosis and antibody-dependent cellular cytotoxicity (ADCC).
Complement Activation: Initiates the complement system, leading to pathogen lysis or opsonization for enhanced phagocytosis.
Multifaceted Functions of Immunoglobulin Antibodies
Immunoglobulin antibodies orchestrate a series of immune responses to protect the body against pathogens:
- Neutralization: By binding to specific sites on pathogens or toxins, antibodies can block their harmful effects.
- Opsonization: Antibodies coat pathogens, making them more attractive targets for phagocytes, which engulf and destroy them.
- Complement System Activation: The Fc region of certain antibodies can initiate the complement cascade, leading to pathogen lysis or opsonization.
- Immobilization and Prevention of Pathogen Adherence: Binding to bacterial flagella or pili can immobilize microbes and prevent them from adhering to host cells.
- Cross-linking (Agglutination): Antibodies can bind to antigens on the surface of pathogens, linking them together and facilitating their removal by phagocytes.
- Antibody-dependent Cellular Cytotoxicity (ADCC): Antibodies can target cells for destruction by natural killer (NK) cells.
Comprehensive Classification of Immunoglobulin Antibodies
Immunoglobulin antibodies are categorized into five primary classes, each with distinct structural and functional characteristics:
1. IgG
- Features: IgG antibodies are the most abundant in the circulatory system, providing long-term protection due to their ability to cross the placenta and offer passive immunity to the fetus.
- Subclasses: IgG is further divided into four subclasses (IgG1, IgG2, IgG3, and IgG4) based on the variability in their heavy chains, influencing their functional properties.
2. IgM
- Features: IgM is the first antibody produced in response to an antigen and is primarily found in the blood and lymphatic fluid. Its pentameric structure allows for effective agglutination and complement activation.
3. IgA
- Features: IgA antibodies are predominantly found in mucosal areas, such as the digestive and respiratory tracts, playing a crucial role in mucosal immunity. They exist in two forms: serum IgA (monomeric) and secretory IgA (dimeric).
4. IgE
- Features: IgE antibodies are involved in allergic reactions and protection against parasitic infections. They bind to mast cells and basophils, triggering the release of histamine and other mediators.
5. IgD
- Features: IgD antibodies are less understood but are believed to play a role in the initiation of immune responses by serving as receptors on B cells.
Table 1: Characteristics of Immunoglobulin Classes
| Class | Heavy Chain | Location | Function |
| IgG | γ | Blood, extracellular fluid | Neutralizes pathogens, opsonization, complement activation |
| IgM | μ | B cell surface, plasma | Primary response to infection, complement activation |
| IgA | α | Mucosal areas, secretions | Barrier protection, neutralization |
| IgE | ε | Bound to mast cells and basophils | Allergic reactions, antiparasitic defense |
| IgD | δ | B cell surface | B cell receptor |
Conclusion
References
- Janeway, C.A., Travers, P., Walport, M., & Shlomchik, M.J. (2016). Immunobiology: The Immune System in Health and Disease. 9th ed. New York: Garland Science.
- Murphy, K., Weaver, C. (2017). Janeway's Immunobiology. 9th ed. New York: Garland Science.
- Abbas, A.K., Lichtman, A.H., & Pillai, S. (2020). Cellular and Molecular Immunology. 10th ed. Philadelphia: Elsevier.
- Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., & Walter, P. (2014). Molecular Biology of the Cell. 6th ed. New York: Garland Science.
- Schroeder, H.W., & Cavacini, L. (2010). Structure and function of immunoglobulins. Journal of Allergy and Clinical Immunology, 125(2 Suppl 2), S41-S52.
- Vidarsson, G., Dekkers, G., & Rispens, T. (2014). IgG subclasses and allotypes: from structure to effector functions. Frontiers in Immunology, 5, 520.
- Woof, J.M., & Burton, D.R. (2004). Human antibody-Fc receptor interactions illuminated by crystal structures. Nature Reviews Immunology, 4(2), 89-99.
- Nimmerjahn, F., & Ravetch, J.V. (2008). Fcγ receptors as regulators of immune responses. Nature Reviews Immunology, 8(1), 34-47.
Written by Zainab Riaz
Zainab Riaz completed her Master degree in Zoology from Fatimah Jinnah University in Pakistan and is currently pursuing a Doctor of Philosophy in Zoology at University of Lahore in Pakistan.
Recent Posts
-
Metabolic Exhaustion: How Mitochondrial Dysfunction Sabotages CAR-T Cell Therapy in Solid Tumors
Imagine engineering a patient's own immune cells into precision-guided missiles against cancer—cells …8th Dec 2025 -
The Powerhouse of Immunity: How Mitochondrial Fitness Fuels the Fight Against Cancer
Why do powerful cancer immunotherapies work wonders for some patients but fail for others? The answe …5th Dec 2025 -
How Cancer Cells Hijack Immune Defenses Through Mitochondrial Transfer
Imagine a battlefield where the enemy doesn't just hide from soldiers—it actively sabotages their we …5th Dec 2025